
Associate Consultant in Haematology
and Oncology, Prince of Wales Hospital
Hodgkin’s lymphoma (HL) is a rare monoclonal lymphoid neoplasm that consists of two subtypes: classical HL (cHL- 95% cases) and nodular lymphocyte predominant HL (NLP-HL)1,2. Notably, cHL has a bimodal age distribution that initially peaks at age 20-30 years, followed by a subsequent peak between the age 50-70 years3. In addition, the incidence of HL in Hong Kong has increased substantially over the last 2-3 years4. Remarkably, the treatment paradigm of HL has evolved over the years, making HL a neoplasm with excellent prognosis5. To understand the evolution of treatment dogma of HL, we have invited Dr. Tommy Tam to share his insight into the local real-world practice.
HL is a rare monoclonal lymphoid neoplasm characterised by the proliferation of large multinucleated cells, also known as Hodgkin and Reed-sternberg (HRS) cells2,5. The incidence of HL in Europe was reported to be around 2-3 per 100,000 individuals5, while in Hong Kong it fluctuates between 0.6 and 1.2 according to the Hong Kong Cancer Registry, with 81 cases registered in 20216. However, recently published statistics suggested an increase of 6.23% in male cases and a 5.39% increase in cases among females, while the mortality rate in both genders decreased since 20224. Despite these reported findings, Dr. Tam suggested that he did not notice an uptrend in the incidence of HL in his clinical practice. Nevertheless, he explained that the rising incidence may partially be driven by the growing numbers of high-risk individuals (smokers, obesity, hypertension and Epstein-Barr virus [EBV] infection). Moreover, with an increased sensitivity in medical imaging, cases are diagnosed more accurately, thereby contributing to the uptrend of HL incidence.
Patients with cHL, especially in young population, commonly present with persistent, painless superdiaphragmatic lymphadenopathy in neck or supraclavicular fossa. Lymphadenopathy may develop within the mediastinum, which triggers symptoms such as cough, substernal chest pain, or anterior chest wall swelling7. Aside from these localised symptoms, HL systemic manifestations include night sweat, unexplained weight loss and recurring fever7, which are similar in both younger and older populations. Because of these noticeable symptoms, patients usually seek medical consultation promptly, thus making timely diagnosis possible. However, in cases where swollen lymph node is the only noticeable abnormality, a rare misjudgement may occur, Dr. Tam added.
Treatment consideration plays an important role in improving treatment tolerability, optimising treatment outcomes, and facilitating the return to a normal life8. The prime concern is the patient’s background, particularly the comorbidities such as the cardiovascular function, history of chronic disease and frailty, according to Dr. Tam. Moreover, it is of paramount importance to choose a treatment algorithm with promising efficacy and safety profile, since treatment tolerability and quality of life play a vital role in patients’ recovery. In addition, consolidation radiotherapy should be used with caution due to an increased risk of breast cancer in female patients who have previously undergone radiotherapy for HL9.
With the emergence of various classes of treatment, the breadth of the landscape in advanced cHL treatment has offered physicians and patients options to minimise adverse effects and optimise treatment efficacy8. Currently, doxorubicin, bleomycin, vinblastine and dacarbazine (ABVD) has replaced escalated bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone (eBEACOPP) to become the most widely adopted treatment regimen in HL10; however, the long-term toxicity observed in HL survivors remains a pressing issue, and some commonly reported adverse effects include pulmonary toxicity, cardiac dysfunction, and secondary malignancies11. Therefore, there is room for improvement by minimising treatment toxicities in the conventional treatment algorithm. Dr. Tam explained that after patients have undergone a few cycles of ABVD, they can be assessed by the positron emission tomography (PET). If response is observed, doses can be lowered, or treatment cycles can be reduced. Specifically, given the well-documented toxicity of bleomycin, the risk of toxicity can be mitigated by removing bleomycin from the combination therapy if patients achieve complete metabolic response upon interim PET scan after 2 cycles of ABVD (iPET2 CMR), as per recent study findings11–13.
Despite these innovations, barriers to improving the treatment outcomes are still to be overcome. Dr. Tam elaborated on the two main barriers to optimising the treatment outcome: financial difficulty and the elimination of chemotherapeutic agents from the treatment protocol. As we are aware that not everyone has a full medical insurance coverage, the affordability issue may hinder their ability to utilise the newer treatment. In addition, some patients are required to fulfil certain eligibility criteria in order to be covered by the government’s subsidy, such as the use of medication in a later line setting. In terms of treatment, Dr. Tam emphasised that the journey to move beyond the reliance on chemotherapy as a mainstream treatment still lies ahead, but the medical community is already moving in the right direction.
As highlighted, the treatment paradigm has been shifted to prioritise safety and tolerability, owing to the toxicity of chemotherapy11. Dr. Tam reiterated that for stage III/IV patients, target therapy is often added to replace the chemotherapy. For instance, anti-cluster of differentiation 30 (anti-CD30) antibody-drug conjugates and anti-programmed cell death protein 1 (anti-PD-1) agents are increasingly prescribed, albeit primarily in the 2nd or 3rd line settings8. As abundant evidence demonstrating the efficacy of these medications emerges, moving the prescription of these medications to the first line can be anticipated8,11. Thus, brentuximab vedotin + AVD (BV+AVD) are gradually replacing bleomycin with brentuximab vedotin (BV) as this may lower the incidence of pulmonary toxicity while improving the efficacy as compared to ABVD regimen14.
The ECHELON-1 study, an open-label, multicentred, randomised phase 3 trial included 1,334 patients in which 664 and 670 patients were randomly assigned to BV+AVD and ABVD groups, respectively14. The primary endpoint of ECHELON-1 study was modified progression free survival (PFS), defined as time to disease progression, death, or modified progression (with the latter defined as evidence of noncomplete response after completion of frontline therapy according to review by an independent committee, followed by subsequent anticancer therapy) and the key secondary end point was overall survival, defined as the time from randomisation14. At a median follow-up of 24.6 months, the 2-year modified PFS in BV+ AVD and ABVD groups were 82.1% (95% confidence interval [CI]: 78.8-85.0) and 77.2% (95% CI: 73.7-80.4), respectively, a difference of 4.9 percentage points (hazard ratio for event of progression, death, or modified progression, 0.77; 95% CI: 0.60-0.98; p=0.04)14. Given the prevalence of the adolescents and young adults (AYA) population in advanced cHL and its significant proportion in the ECHELON-1 trial (accounting for 58% of the intend-to-treat [ITT] population), a subgroup analysis of ECHELON-1 was started in the AYA patients aged from 18 to 39 to evaluate the consistency of the safety and efficacy of BV+AVD regimen at 6 years15. Among them, 396 patients received the BV+AVD regimen and 375 received ABVD regimen15 .
Remarkable consistency in the PFS and OS benefits with the ITT population was demonstrated. A staggering 36% risk reduction in disease progression at 6 years was found in the BV+AVD arm compared to those in the ABVD arm, with the 6-year PFS rate of 86.4% with BV+AVD versus 79.4% with ABVD (HR 0.636; 95% CI: 0.445-0.908; P=0.012)(Figure 1)15. The younger patients aged 18-29 years showed a similar outcome, with 6-year PFS rate of 87.3% with BV+AVD and 80.0% with ABVD (HR 0.604; 95% CI: 0.378-0.965; P=0.033)15. Across ages 18 to 39, numerical PFS benefit with BV+AVD versus ABVD regardless of PET2 status was noted. Furthermore, after 71.7 months of median OS follow-up, a striking reduction in death of 60% was discovered, as the estimated 6-year survival rate was 98.2% for BV+AVD arm and 94.9% for the ABVD arm (HR 0.391; 95% CI: 0.161-0.951; P=0.032)15, thus underscoring the sustained efficacy of BV+AVD in disease control and preventing progression across subgroups.
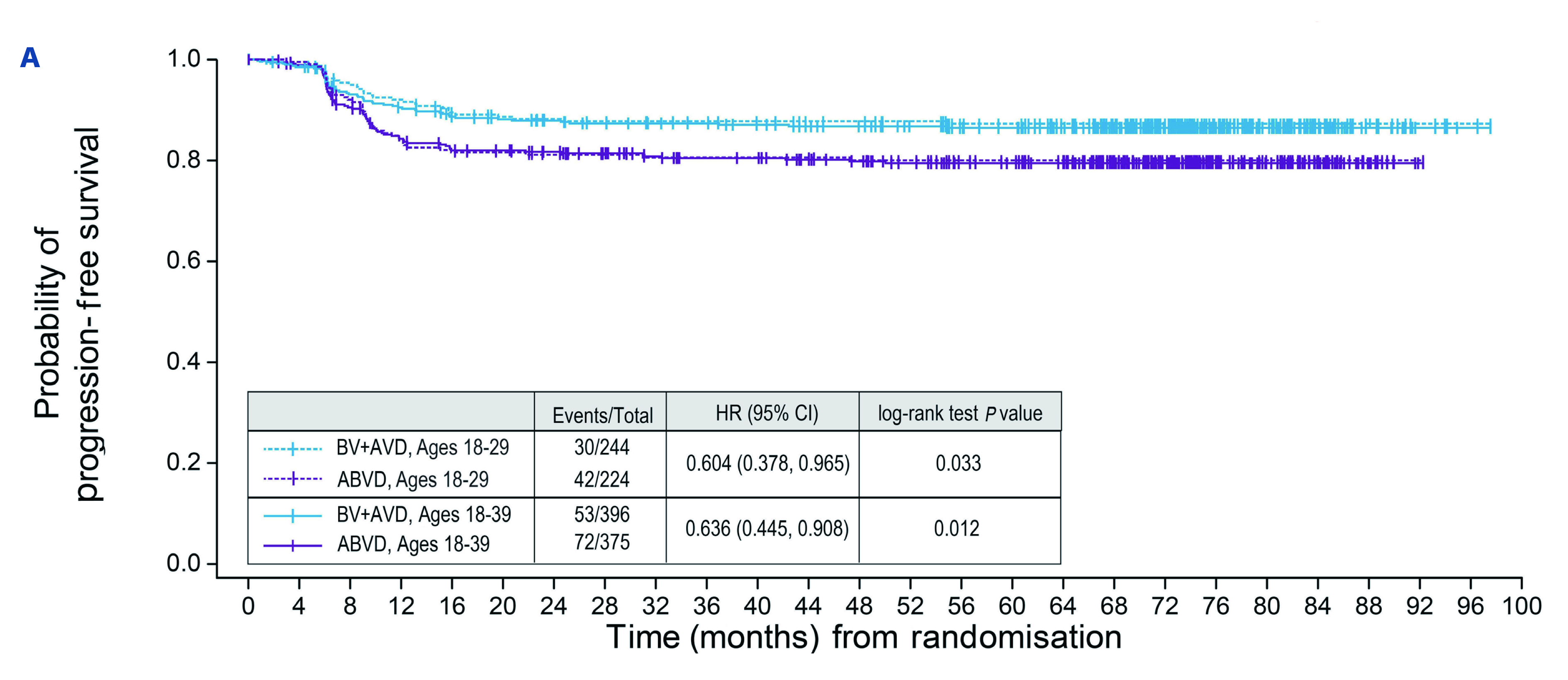
Figure 1. Progression-free survival per investigator since time from randomisation (Adapted from Crosswell H, et al. 2023)15
Since the long-term sequelae of HL treatment have been a concern in long-term survivorship11,16, adverse events have been analysed in this study. Notably, no impact on fertility was seen in females on BV+AVD regimen, in that BV+AVD arm has a higher pregnancy rate of 23.4% compared to 16.8% in the ABVD arm. Nor did males suffer from infertility, as 31 and 30 partners of the male HL patients were able to become pregnant in BV+AVD and ABVD groups, respectively. As for secondary malignancies, less than 2% of second cancers were found in each arm, which was even lower than the ITT population in ECHELON-1 trial (3.5% with BV+AVD and 4.9% with ABVD)15,17, thus establishing the long-term safety of BV+AVD regimen. Finally, a higher risk of developing peripheral neuropathy (PN) was identified, with 64% in the BV+AVD arm and 40% in the ABVD arm reported to have any-grade PN. Fortunately, 78% of patients attained a complete resolution and 11% had an improvement at 6 years (Figure 2), which means proactive management makes PN a manageable side effect15.
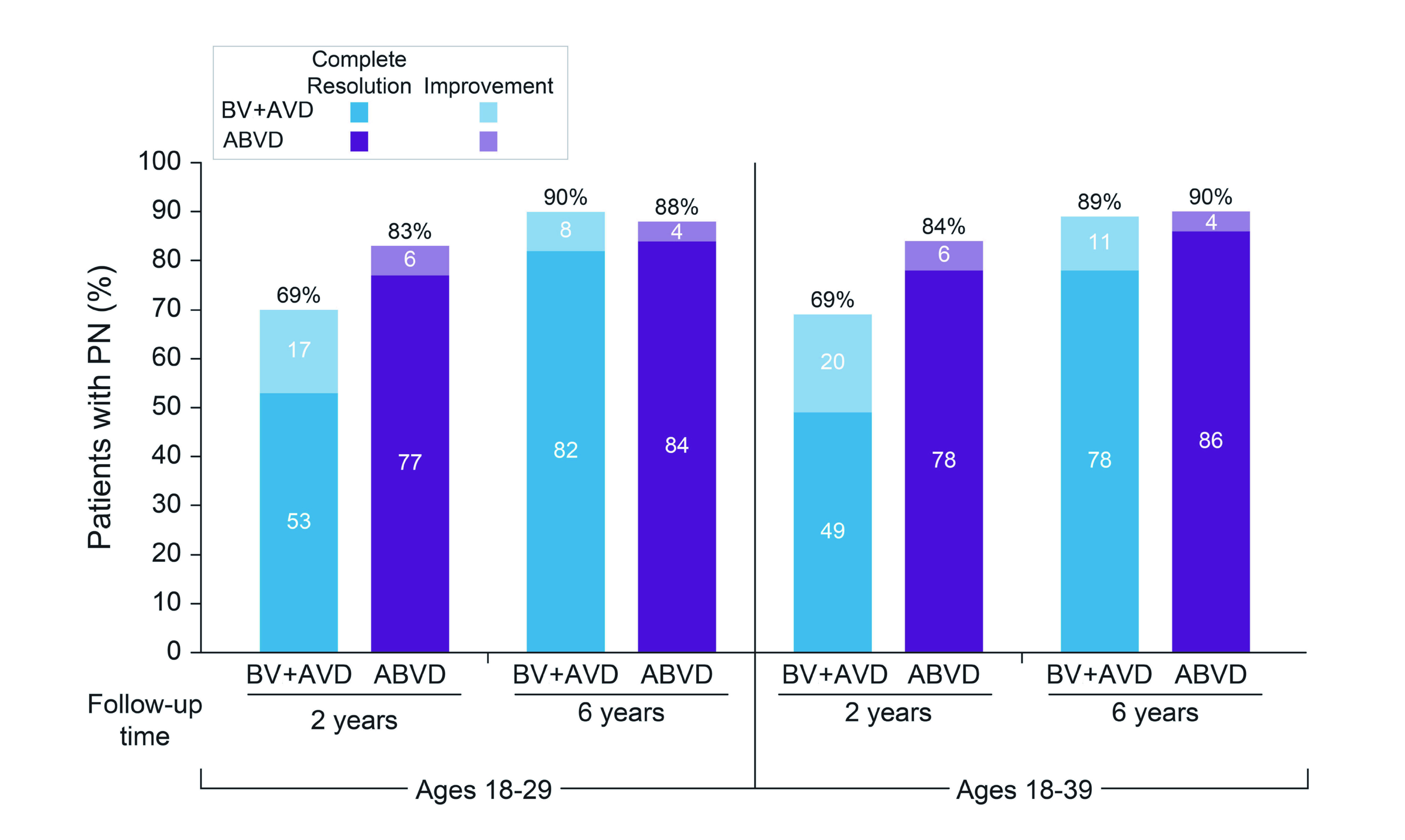
Figure 2. Rate of complete resolution and improvement in peripheral neuropathy analysed at 2 years and 6 years (Adapted from Crosswell H, et al. 2023)15
To illustrate the benefits of BV+AVD regimen in young adults, Dr. Tam shared a case involving a young female diagnosed with stage 3 cHL. The patient initially presented with chest pain and dyspnoea. The PET scan revealed a large tumour in the mediastinum and a few lesions in other parts of the body. Considering that the patient chose to continue a career in performing art, Dr. Tam believed choosing an efficacious regimen with minimal pulmonary toxicity would be of utmost importance. Added that the patient was young at the time of diagnosis, the risks of secondary diseases resulting from chemotherapeutic agents and radiotherapy were avoided. As a result, Dr. Tam opted for 6 cycles of BV+AVD regimen for the patient, and remarkably, a treatment response was observed only after 1-2 cycles as the chest symptoms resolved. After 6 treatment cycles, the patient achieved complete remission, and had been in remission for 3 years.
Patients with advanced cHL are often faced with enormous treatment burden and risk of relapse. Thankfully, an effort to minimise the treatment toxicity burden is witnessed with groundbreaking agents and modified treatment algorithms. Additionally, the international data and clinical experience have laid a solid foundation for refining the treatment protocol of advanced cHL8. BV+AVD has been utilised by local doctors for some years and the medical community has sufficient clinical experience with the variations of this regimen, according to Dr. Tam. Nevertheless, to maximise the therapeutic benefits of advanced HL treatment, doctors could pay closer attention to the signs exhibited by patients and make prompt dosage adjustments if necessary. BV+AVD regimen showed a superior efficacy compared to the ABVD regimen in treating patients with advanced cHL14. As the new era of cHL treatment ushers a brighter treatment prospectus, it is merely a matter of time for advanced cHL to be treated like a chronic disease.
References
1. Hanel W, Herrera AF, Epperla N. Management of classical Hodgkin lymphoma: a look at up to date evidence and current treatment approaches. Exp Hematol Oncol 2022; 11: 1–13. 2. Kaseb H, Babiker HM. Hodgkin Lymphoma. StatPearls 2023; published online June 26. https://www.ncbi.nlm.nih.gov/books/NBK499969/ (accessed June 17, 2024). 3. Zhou L, Deng Y, Li N, et al. Global, regional, and national burden of Hodgkin lymphoma from 1990 to 2017: estimates from the 2017 Global Burden of Disease study. J Hematol Oncol 2019; 12: 107. 4. Huang J, Pang WS, Lok V, et al. Incidence, mortality, risk factors, and trends for Hodgkin lymphoma: a global data analysis. J Hematol Oncol 2022; 15: 1–11. 5. Connors JM, Cozen W, Steidl C, et al. Hodgkin lymphoma. Nat Rev Dis Primers 2020; 6: 61. 6. Hong Kong Cancer Registry, Hospital Authority. https://www3.ha.org.hk/cancereg/allagesresult.asp (accessed June 6, 2024). 7. Connors JM. Clinical Manifestations and Natural History of Hodgkin’s Lymphoma. The Cancer Journal 2009; 15: 124–8. 8. Mohty R, Dulery R, Bazarbachi AH, et al. Latest advances in the management of classical Hodgkin lymphoma: the era of novel therapies. Blood Cancer J 2021; 11: 126. 9. De Bruin ML, Sparidans J, van’t Veer MB, et al. Breast Cancer Risk in Female Survivors of Hodgkin’s Lymphoma: Lower Risk After Smaller Radiation Volumes. Journal of Clinical Oncology 2009; 27: 4239–46. 10. Vellemans H, André MPE. Review of Treatment Options for the Management of Advanced Stage Hodgkin Lymphoma. Cancers (Basel) 2021; 13: 3745. 11. Follows AM, Santarsieri A. Minimising the Toxicities of First Line Hodgkin Lymphoma Treatment in the Modern Era. Cancers (Basel) 2022; 14: 5390. 12. Eichenauer DA, Aleman BMP, André M, et al. Hodgkin lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology 2018; 29: iv19–29. 13. Johnson P, Federico M, Kirkwood A, et al. Adapted Treatment Guided by Interim PET-CT Scan in Advanced Hodgkin’s Lymphoma. New England Journal of Medicine 2016; 374: 2419–29. 14. Connors JM, Jurczak W, Straus DJ, et al. Brentuximab Vedotin with Chemotherapy for Stage III or IV Hodgkin’s Lymphoma. New England Journal of Medicine 2018; 378: 331–44. 15. Crosswell HE, LaCasce AS, Bartlett NL, et al. Brentuximab vedotin with chemotherapy in adolescents and young adults with stage III or IV classical Hodgkin lymphoma in ECHELON-1. Haematologica 2023; published online Oct 5. DOI:10.3324/haematol.2023.283303. 16. Nassi L, De Sanctis V, Loseto G, et al. Second Cancers in Classical Hodgkin Lymphoma and Diffuse Large B-Cell Lymphoma: A Systematic Review by the Fondazione Italiana Linfomi. Cancers (Basel) 2022; 14: 519. 17.Ansell SM, Radford J, Connors JM, et al. Overall Survival with Brentuximab Vedotin in Stage III or IV Hodgkin's Lymphoma. New England Journal of Medicine 2022; 387(4): 310-20.





