
Senior Consultant Haematologist and Transplant Physician
Department of Haematology
SingHealth Duke-NUS Blood Cancer Center,
Singapore General Hospital and National Cancer,
Singapore
Follicular lymphoma (FL) is an indolent form of non-Hodgkin’s lymphoma (NHL) that originates from germinal centre B cells1 and is the second most common NHL subtype worldwide. Although first-line treatment achieves a complete response (CR) and sustained remission in most patients with FL, a substantial proportion of the patients experience serial relapses, become refractory to treatment, or develop histologic transformation to aggressive B-cell lymphoma2. Therefore, new therapies with promising efficacy and safety profile are highly desirable. While Obinutuzumab-based treatment has been reported to yield survival benefits in patients with FL3, recent trials have demonstrated that the combined treatment of Zanubrutinib and Obinutuzumab results in further improved progression-free survival (PFS), with a preferable tolerability2. In a recent sharing, Dr. Chandramouli Nagarajan presented the clinical findings of Zanubrutinib-based treatment and discussed its potential role in managing FL.
FL is one of the most common indolent NHL subtypes in Western countries. For instance, Gupta et al. (2022) reported that the incidence of FL reached 3.18 cases per 100,000 people annually in the United States, accounting for approximately 20-25% of all NHLs1. Dr. Nagarajan highlighted that FL is more prevalent among older adults, with the median age of FL patients being 65 years old. Moreover, FL appears to occur more frequently in male patients, with a male-to-female ratio of 1.2:11. Notably, differences in incidence rates for FL by ethnicity were observed, with the highest rates in Caucasians, which suggests potential genetic differences in susceptibility to the disease4.
FL is a biologically heterogeneous disease with a diverse range of clinical presentations. However, most patients present with an indolent disease course, with continuous patterns of relapse and progressively shorter disease-control intervals with each line of treatment5. Although FL is still considered incurable, the life expectancy for most patients is long, with a 5-year relative survival rate of 90%6.
The t(14;18) translocation is the genetic hallmark of FL, which gives rise to a BCL2-IGH fusion and is observed in >85% of FL. While the low levels of t(14;18) translocation can be detected in 50-70% of presumably normal adults, higher levels of circulating t(14;18) cells, greater than 1 in 10,000 blood cells, are associated with a 23-fold higher risk of developing FL4. Remarkably, components of the B-cell receptor signalling pathway, such as phosphoinositide 3-kinase (PI3K), Bruton tyrosine kinase (BTK), and spleen tyrosine kinase, are frequently activated in FL5, suggesting the potential application of kinase inhibitors for treating FL.
"Treatment of FL is primarily based on symptoms rather than stage or biology", Dr. Nagarajan addressed. Indeed, existing literature advocates that FL treatment aims to decrease symptom burden and morbidity, improve quality of life, and prolong survival1. Regarding relapsed or refractory (R/R) FL, there is currently no standard management for the condition. Nonetheless, many drug combinations, novel agents, and transplant options have been studied and have demonstrated efficacy.
Practically, the efficacy of anti-CD20 monoclonal antibody has been established. However, the choice of chemotherapy backbone is still controversial. Accordingly, the FOLL05 trial by Luminari et al. compared the clinical outcomes achieved by various chemoimmunotherapy (CIT) regimens, including R-CVP (rituximab plus cyclophosphamide, vincristine, and prednisone), R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone), and R-FM (rituximab plus fludarabine and mitoxantrone), in treatment-naïve patients with advanced FL7.
Based on a median of 7 years follow-up of the FOLL05 trial, the hazard ratio (HR) for PFS adjusted by FL International Prognostic Index 2 versus R-CVP was 0.73 for R-CHOP (p=0.037) and 0.67 for R-FM (p=0.009, Figure 1). The 8-year overall survival (OS) rate was 83%, with no significant differences among the study arms7. The results thus confirmed the favourable OS with the CIT regimens, but R-CVP was not as effective as R-CHOP and R-FM in enhancing PFS.
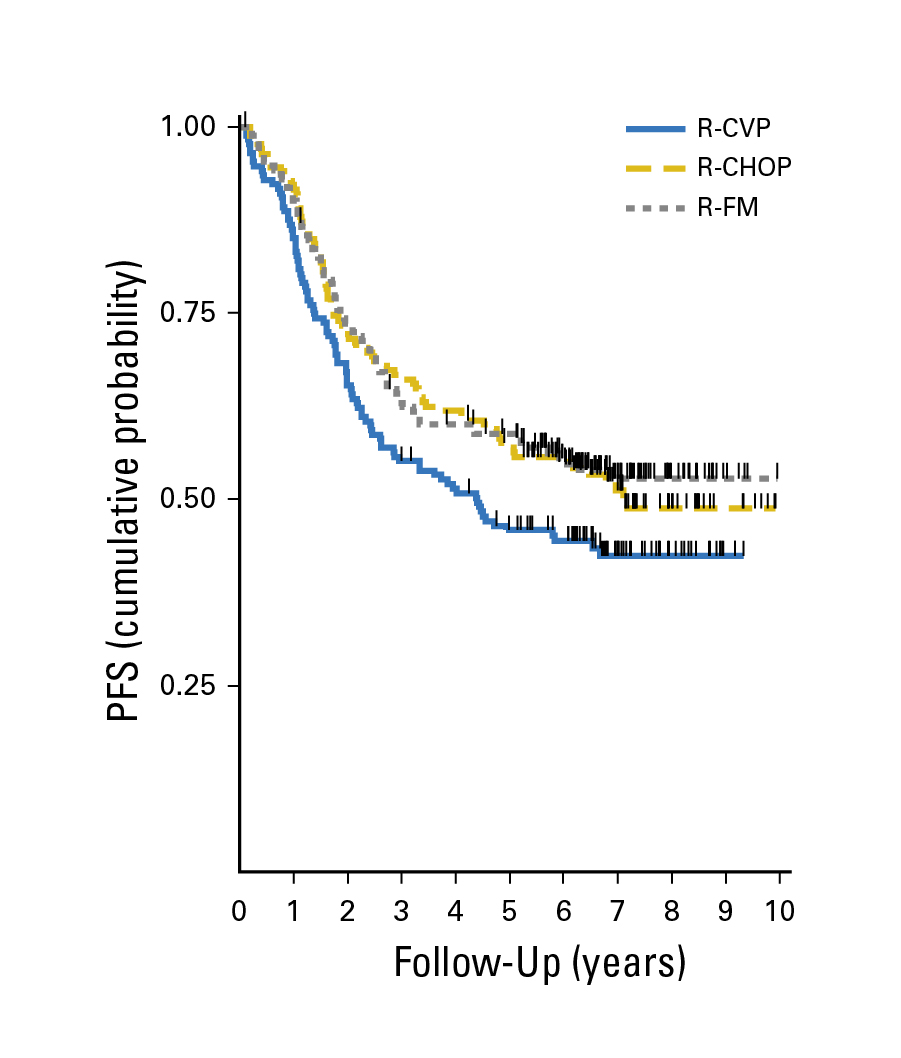
Figure 1: PFS achieved in the FOLL05 trial7
In the StiL trial by Rummel et al. (2013), the efficacy of bendamustine plus rituximab (B-R) as first-line treatment for patients with indolent and mantle-cell lymphomas was compared with that of R-CHOP. At a median follow-up of 45 months, the median PFS was significantly longer in the B-R group than in the R-CHOP group (69.5 months vs 31.2 months, HR: 0.58, p<0.0001, Figure 2). The results also indicated that B-R was better tolerated than R-CHOP8. Hence, B-R would be a preferred first-line treatment approach relative to R-CHOP.
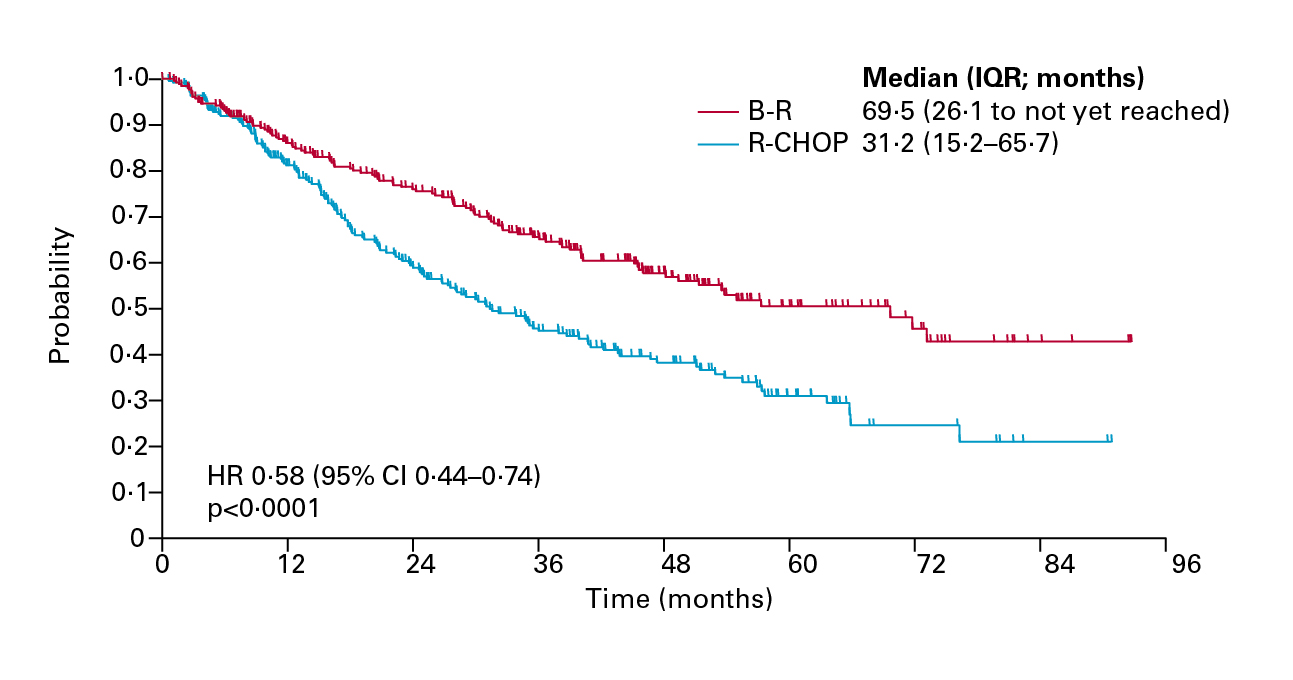
Figure 2. PFS achieved by B-R and R-CHOP8
Subsequently, the clinical performance of B-R, R-CHOP, or R-CVP in treatment-naïve patients with indolent NHL or mantle-cell lymphoma was evaluated in the BRIGHT trial. The 5-year follow-up results revealed that the PFS rates at 5 years were 65.5% in the B-R group, which was significantly higher than those in the R-CHOP/R-CVP group (55.8%, HR: 0.61, p=0.0025, Figure 3). The HR for event-free survival (p=0.002) and duration of response (p=0.0134) also favoured the B-R regimen over R-CHOP/R-CVP. Nonetheless, no significant difference in OS was observed9. The results further supported that B-R provided better long-term disease control than R-CHOP/R-CVP.
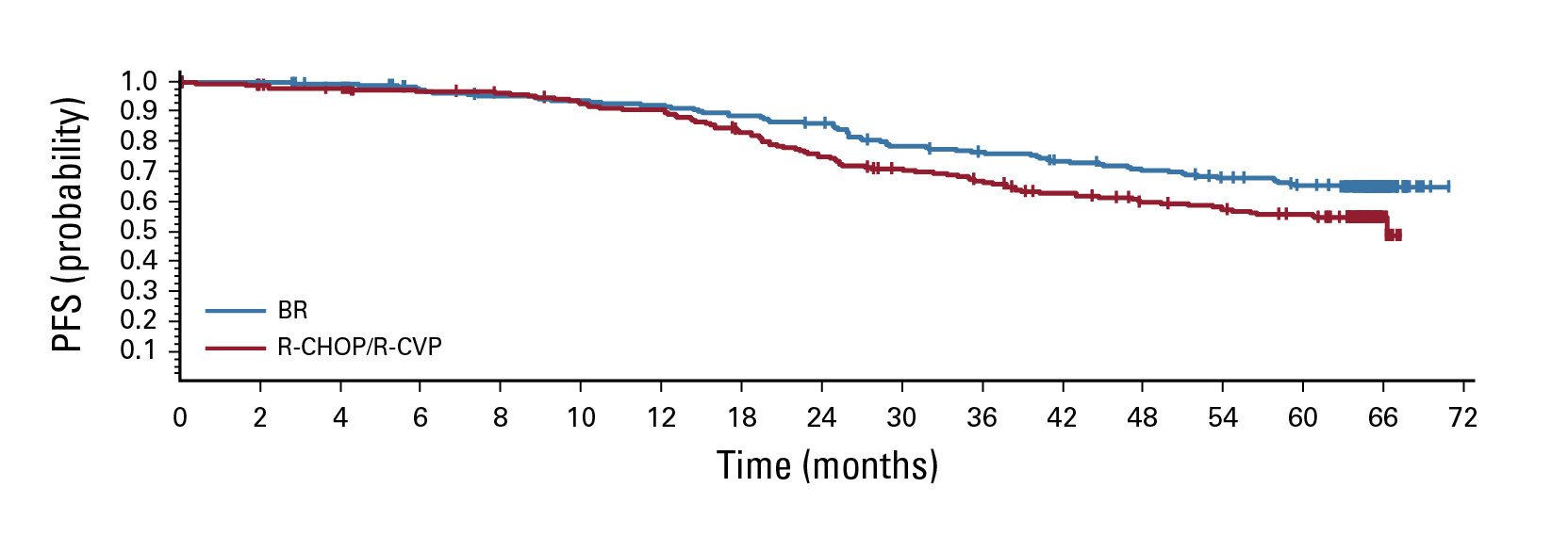
Figure 3. PFS achieved by B-R and R-CHOP/R-CVP9
According to the ESMO clinical guidelines, patients with FL are classified as either high-tumour burden, who are usually treated with CIT with or without maintenance, or low-tumour burden, for whom a watch and wait (WW) strategy is recommended until disease progression10. Nonetheless, Dr. Nagarajan highlighted the unmet need in FL that about 20% of patients with FL experience disease progression within 2 years of first-line treatment, known as POD24, and consequently have an unfavourable prognosis. It has been reported that the 5-year OS of FL patients experiencing POD24 was 50%, whereas that of the non-POD24 group was 90%11.
Dr. Nagarajan further addressed that POD24 is a post-treatment endpoint and, therefore, cannot be used to guide upfront treatment decisions. Given that POD24 can be evaluated based on biopsies, it can serve as a tool for identifying high-risk FL patients and should be routinely considered11.
While biopsy at relapse is essential for ruling out transformation to aggressive histology, prescribing more intensive or novel therapeutic strategies may be beneficial for improving the survival and quality of life (QoL) of patients with R/R FL. In recent years, Bruton tyrosine kinase inhibitors (BTKi) have emerged as promising therapeutic options for R/R FL. Particularly, Zanubrutinib is a potent, specific, and irreversible second-generation covalent BTKi, which achieves deep and durable responses in patients with various B-cell malignancies12.
The efficacy of Zanubrutinib monotherapy in countering R/R FL was examined in the phase 1/2, open-label, multicentre, single-agent trial by Phillips et al. (2022), which involved 20 R/R marginal zone lymphoma (MZL) and 33 R/R FL patients. In patients with R/R FL, the overall response rate (ORR) was 36.4%, and the complete response (CR) rate was 18.2%. After a median follow-up of 33.9 months, the median PFS was 10.4 months. The treatment with Zanubrutinib was generally well tolerated, with most adverse events being grade 2 or lower12. The results suggested a favourable benefit-to-risk profile of Zanubrutinib, which could be a promising addition to available therapies for patients with R/R FL.
To illustrate the efficacy of Zanubrutinib in combined therapies against R/R FL, Dr. Nagarajan presented the findings in the ROSEWOOD trial. In the trial, patients with R/R FL who had received at least 2 lines of therapy were randomly assigned at a 2:1 ratio to receive either the combined therapy of Zanubrutinib and Obinutuzumab (ZO), an anti-CD20 therapy, or Obinutuzumab (Ob) monotherapy2.
After a median follow-up of 20.2 months, the ORR by independent central review (ICR) achieved with ZO treatment (n=145) was 69%, which was significantly higher than that of Ob (n=72, ORR=46%, p=0.001). Of note, the ORR advantage of ZO over Ob did not differ substantially across prespecified subgroups, such as gender, previous lines of therapy, FLIPI risk category, etc. Moreover, the CR rate of the ZO group was 39%, whereas that of Ob was 19% (p=0.004). The partial response (PR) rates for the ZO and Ob groups were 30% and 26%, respectively. Importantly, the 18-month duration of response (DOR) rate was 69% in the ZO group and 42% in the Ob group (Figure 4)2.
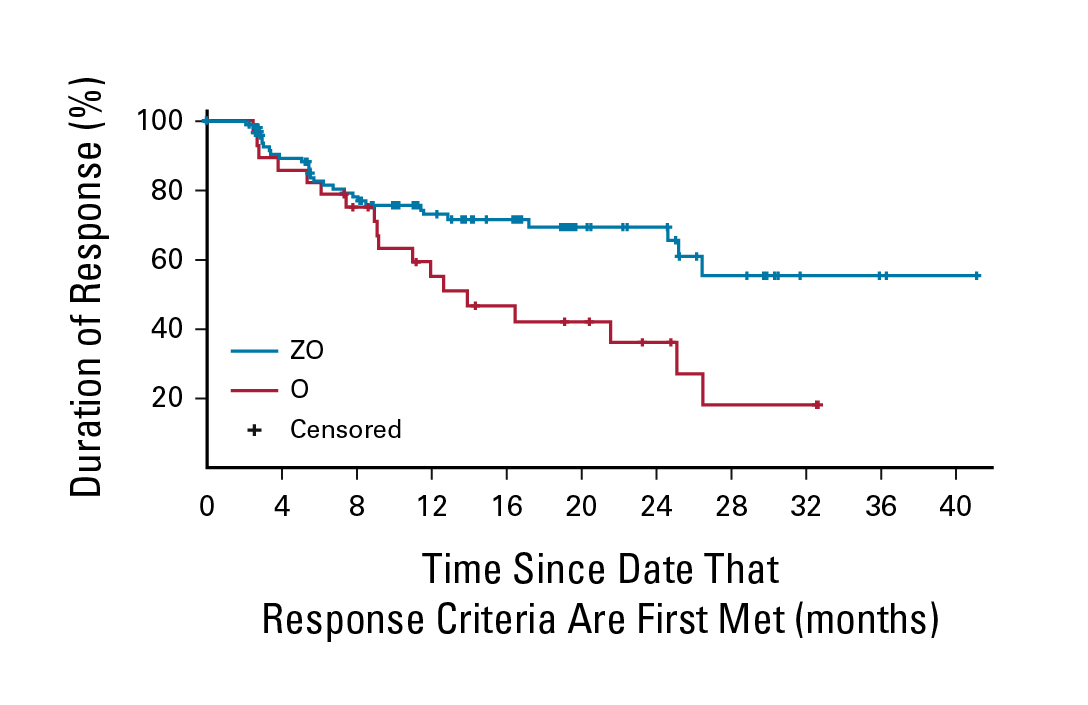
Figure 4. DOR response per ICR by treatment2
Regarding survival benefits, the estimated OS rate at 24 months was numerically higher (p=0.085) with ZO (77%) compared to Ob (71%). However, the median PFS of the ZO group (28.0 months) was significantly longer than that of Ob (10.4 months, HR: 0.50, p<0.001, Figure 5). In particular, the time to next treatment (TTNT) was not estimable with ZO, which was significantly longer than that achieved with Ob (12.2 months, HR: 0.34, p<0.001, Figure 6). Furthermore, the ZO toxicity profile is consistent with the known and tolerable safety profiles of each drug, with no unexpected additional concerns2. Based on the results, the ZO treatment demonstrated meaningful efficacy and a manageable safety profile in heavily pretreated patients with R/R FL.
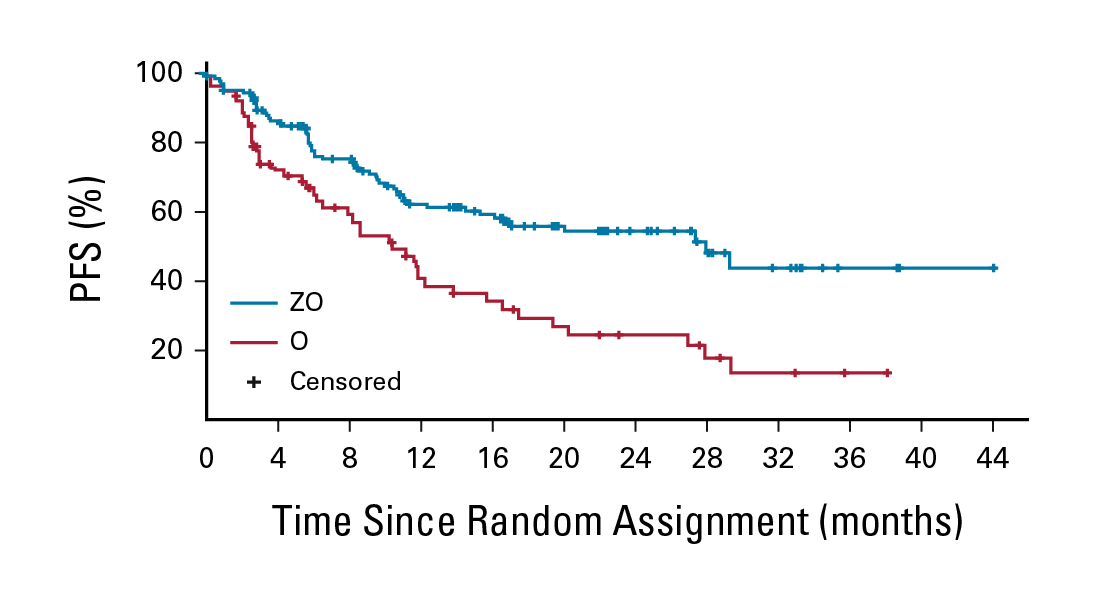
Figure 5. PFS per ICR by treatment2
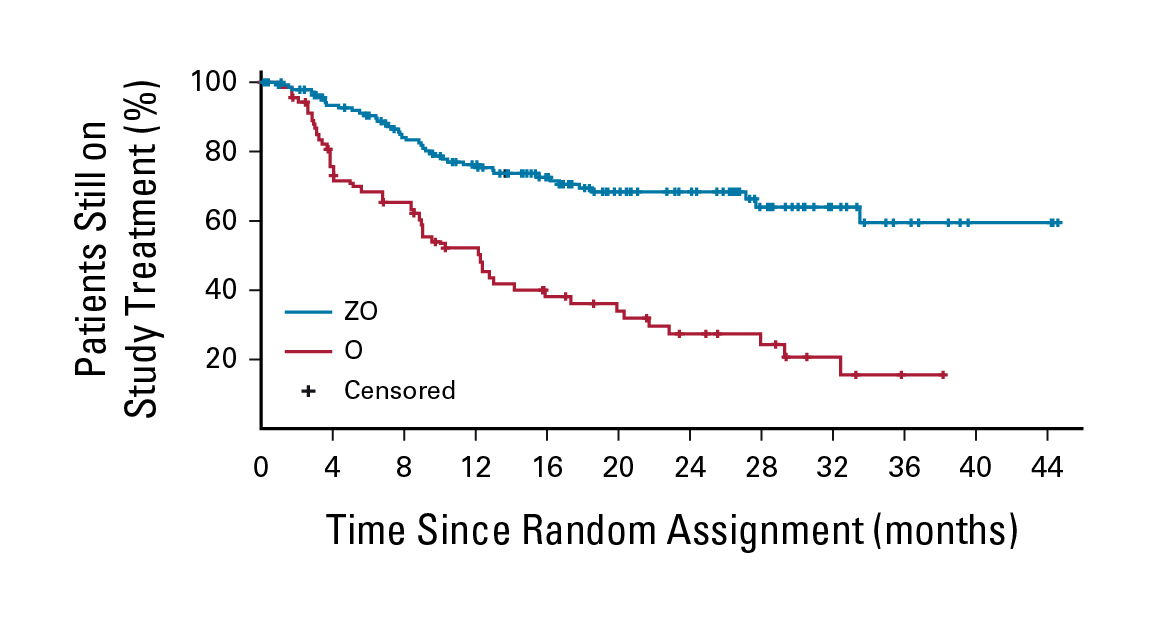
Figure 6. TTNT achieved by ZO and Ob2
Recent developments in therapeutics for FL have substantially improved the outcomes for most patients, with survival often lasting a decade or more, and many are functionally cured. Nonetheless, the development of relapsed or refractory disease is still a clinical challenge. As per Dr. Nagarajan, biopsy at relapse is crucial in identifying high-risk FL patients, whereas timely prescription of novel therapeutic strategies lead to better patient outcomes. "An understanding of the patient profiles is important for optimising the individualised therapy," he emphasised.
In view of the results of ROSEWOOD trial, Dr. Nagarajan noted that BTKi is valuable across different low-grade NHLs in combination with anti-CD20 therapies. In addition to BTKis, he looked forward to witnessing the development of newer-generation immunotherapies and the therapeutic benefits they brought to patients with FL.
References
1. Gupta G, Garg V, Mallick S, Gogia A. Current trends in diagnosis and management of follicular lymphoma. Am J Blood Res 2022; 12: 105. 2. Zinzani PL, Mayer J, Flowers CR, et al. ROSEWOOD: A Phase II Randomized Study of Zanubrutinib Plus Obinutuzumab Versus Obinutuzumab Monotherapy in Patients with Relapsed or Refractory Follicular Lymphoma. Journal of Clinical Oncology 2023; 41: 5107–17. 3. Townsend W, Hiddemann W, Buske C, et al. Obinutuzumab Versus Rituximab Immunochemotherapy in Previously Untreated iNHL: Final Results From the GALLIUM Study. Hemasphere 2023; 7: E919. 4. Cerhan JR. Epidemiology of Follicular Lymphoma. Hematol Oncol Clin North Am 2020; 34: 631. 5. Zinzani PL, Muñoz J, Trotman J. Current and future therapies for follicular lymphoma. Exp Hematol Oncol 2024; 13: 1–11. 6. American Cancer Society. Survival Rates and Factors That Affect Prognosis (Outlook) for Non-Hodgkin Lymphoma. 2024. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/factors-prognosis.html (accessed June 4, 2025). 7. Luminari S, Ferrari A, Manni M, et al. Long-Term results of the FOLL05 trial comparing R-CVP Versus R-CHOP versus R-FM for the initial treatment of patients with advanced-stage symptomatic follicular lymphoma. Journal of Clinical Oncology 2018; 36: 689–96. 8. Rummel MJ, Niederle N, Maschmeyer G, et al. Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: An open-label, multicentre, randomised, phase 3 non-inferiority trial. The Lancet 2013; 381: 1203–10. 9. Flinn IW, Van Der Jagt R, Kahl B, et al. First-Line Treatment of Patients With Indolent Non-Hodgkin Lymphoma or Mantle-Cell Lymphoma With Bendamustine Plus Rituximab Versus R-CHOP or R-CVP: Results of the BRIGHT 5-Year Follow-Up Study. Journal of Clinical Oncology 2019; 37: 984. 10. Dreyling M, Ghielmini M, Rule S, et al. Newly diagnosed and relapsed follicular lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology 2021; 32: 298–308. 11. Leonard JP. POD24 in follicular lymphoma: time to be “wise”. Blood 2022; 139: 1609–10. 12. Phillips T, Chan H, Tam CS, et al. Zanubrutinib monotherapy in relapsed/refractory indolent non-Hodgkin lymphoma. Blood Adv 2022; 6: 3472–9.





