

Specialist in Cardiology
Honorary Clinical Professor
Department of Medicine
The University of Hong Kong
Since the publication of the 2019 European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS) Guidelines for the management of dyslipidemia, various landmark clinical trials regarding therapies for dyslipidemia have been published. Accordingly, the ESC Clinical Practice Guidelines Committee has recently announced the 2025 Focused Update, which addresses the changes in recommendations for the treatment of dyslipidemia based on the newly reported evidence1. To highlight the clinical implications of the updated guidelines, Prof. Lau Chu Pak was invited to share his insights into the key updates. Particularly, Prof. Lau also discussed the roles of statin-based combined therapies in optimizing dyslipidemia management.
The 2025 Focused Update has incorporated the findings of several randomized controlled trials (RCTs) that might change patient management ahead of the next scheduled full dyslipidemia guidelines. Prof. Lau outlined that the explicit classification of the “Extreme risk” category on top of the existing “Very high risk” is a key focus of the updated guidelines. Patients with atherosclerotic cardiovascular disease (ASCVD) who experience recurrent vascular events while taking maximally tolerated statin-based therapy, or those who have polyvascular arterial disease are classified as having an extreme risk of cardiovascular (CV) events1.
Prof. Lau addressed that the updated risk classification precisely indicates the recommended treatment target for low-density lipoprotein cholesterol (LDL-C) for each risk category (Figure 1)1, hence guiding the appropriate treatments. “Patients with documented ASCVD, such as with significant plaque on coronary computed tomography (CT) scan, are classified as “Very high risk” under the updated guidelines, and the recommended treatment target for LDL-C is <1.4 mmol/L and ≥50% reduction from baseline,” he noted. Remarkably, the treatment target for LDL-C is <1.0 mmol/L and ≥50% reduction from baseline for the Extreme risk category1.
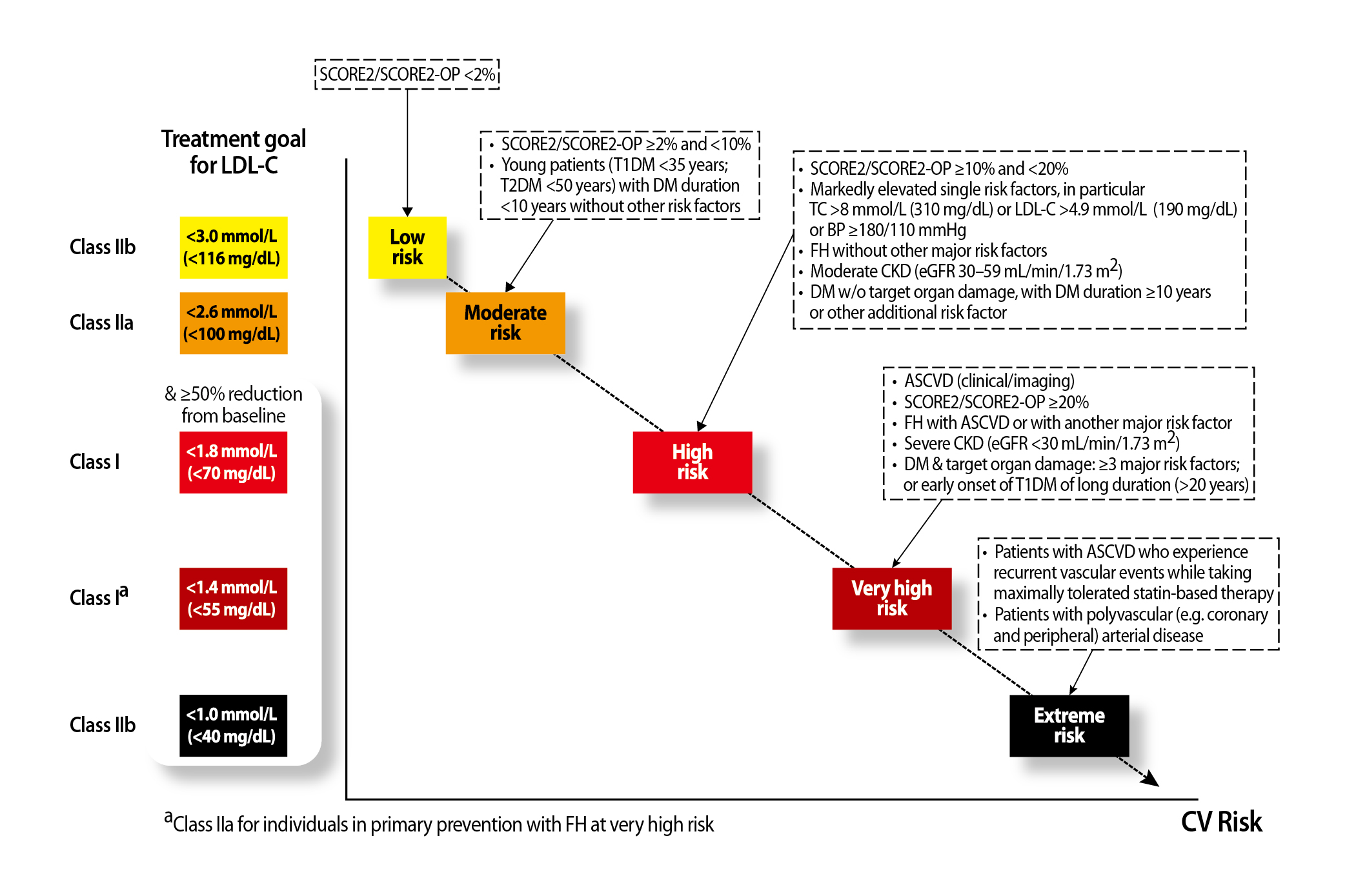
Figure 1: Treatment goals for LDL-C across categories of total CV risk1, BP: blood pressure; CKD: chronic kidney disease; DM: diabetes mellitus; eGFR: estimated glomerular filtration rate; FH: familial hypercholesterolemia; SCORE2: Systematic Coronary Risk Evaluation 2; SCORE2-OP: Systematic Coronary Risk Evaluation 2-Older Persons; TC: total cholesterol level
By virtue of the aggressive LDL-C goal for patients at extreme risk, Prof. Lau stated that most of these patients are already on statin therapy and thus will require add-on medications to achieve the treatment target. Of note, the reduction in the risk of CV events with ezetimibe in the absence of statin therapy in patients aged ≥75 years and without history of coronary artery disease was demonstrated in the EWTOPIA 75 trial2. In this regard, the use of non-statin therapies has been highlighted in the updated guidelines. For instance, the addition of bempedoic acid (BA) to the maximally tolerated dose of statin, with or without ezetimibe, is recommended in patients at high or very high risk to achieve the LDL-C goal. Prof. Lau quoted that the oral combined treatments would reduce LDL-C by 58-68% (Figure 2)1.
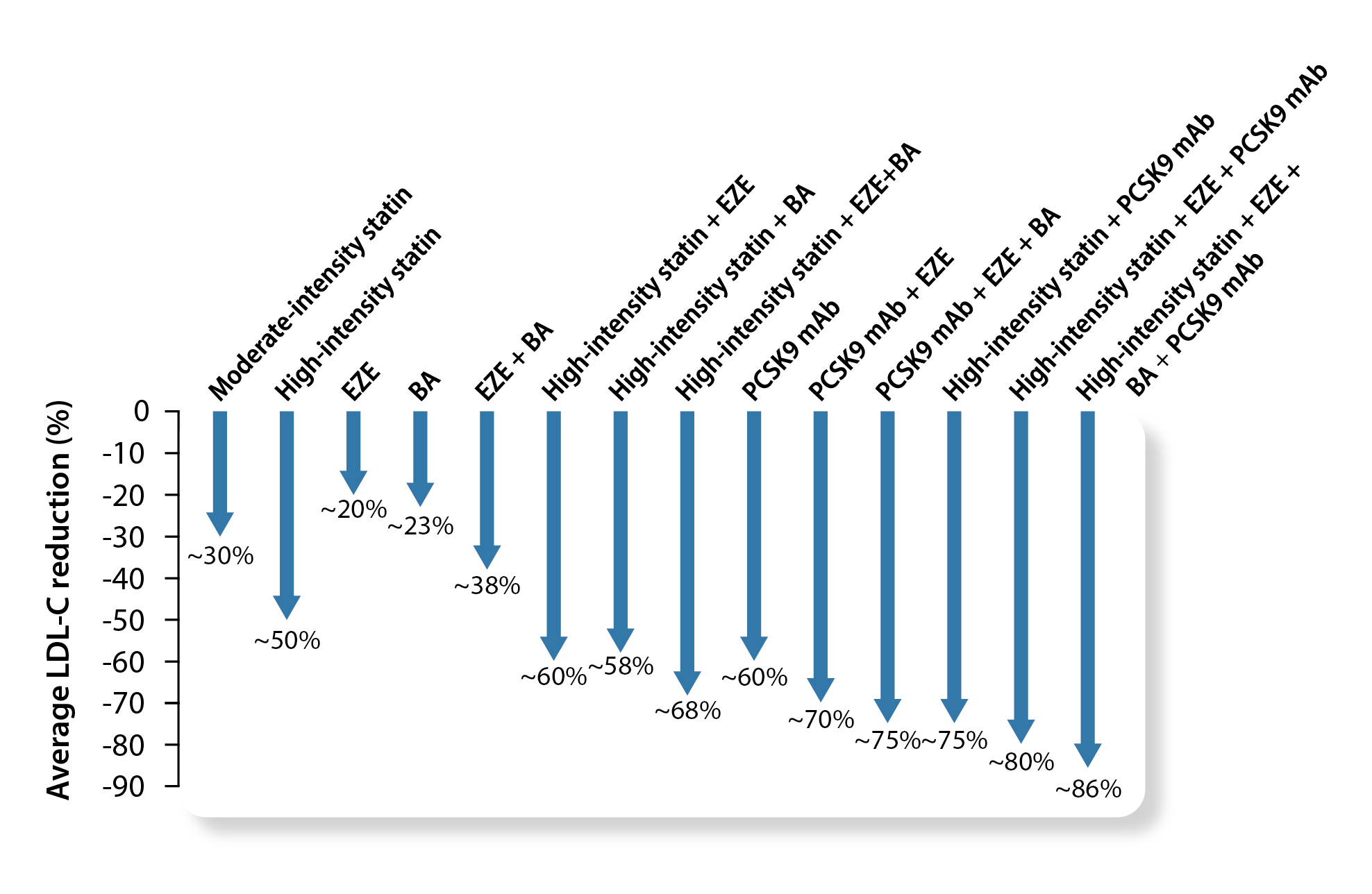
Figure 2: Average reduction in low-density lipoprotein cholesterol levels with different pharmacological therapies1, BA: bempedoic acid, EZE: ezetimibe, PCSK9 mAb: proprotein convertase subtilisin-kexin type 9 monoclonal antibodies
Achieving the recommended LDL-C goals can be clinically challenging. Notably, the reported mean LDL-C reduction with monotherapy of a moderate-intensity statin was 30%, whereas those with ezetimibe and BA monotherapy were only 20% and 23%, respectively1.
In response to the inquiry on first-line therapy for LDL-C lowering, Prof. Lau emphasized that the foremost consideration is whether primary or secondary prevention is required. “A more aggressive treatment approach is needed for secondary prevention to substantially and rapidly reduce the LDL-C level, especially in patients with a high baseline LDL-C level,” he opined. In contrast, a more gradual up-titration in primary prevention.
A former study by Kytö et al. (2022) indicated that patients not using statins early after myocardial infarction (MI) had higher risks of all-cause mortality and major adverse cardiovascular event3. Besides, it has been reported that post-percutaneous coronary intervention (PCI) statin treatment significantly reduced the incidence of cardiac death and recurrent MI compared to those without statins4. Thus, Prof. Lau stressed that combined treatment with the highest tolerable statin and add-on therapy should be initiated as early as possible for patients with MI or those who have undergone PCI.
Apart from MI and PCI, Prof. Lau stressed that the baseline LDL-C of patients has to be considered. If a 40-50% LDL-C reduction is required, combined treatment of moderate-intensity statin and add-on therapy is recommended rather than high-intensity statin due to better tolerability. Indeed, combined moderate-intensity statin and ezetimibe has been reported to provide noninferior efficacy to high-intensity statins for LDL-C reduction and CVD prevention, with a lower risk of side effects and better treatment adherence5.
Furthermore, Prof. Lau highlighted the importance of considering side effect profiles and patient adherence when selecting LDL-C lowering therapies. Of note, different intensities and types of statin therapy may have different muscle side effects, and other side effects like liver dysfunction and gout have to be considered. Most of these side effects are readily reversible6. Practically, injectable medications may be avoided in some patients with needle phobia to achieve the target LDL-C level.
As statin-based treatments are the mainstay of LDL-C control, the comparative efficacy of statin monotherapy and statin-based combined therapies has attracted the interest of researchers and clinicians. Recently, a clinical study using medication prescription data and including 31,993 patients after PCI from a national cohort by Lee et al. (2024) demonstrated that combined therapy of moderate-intensity atorvastatin and ezetimibe was associated with a significantly lower incidence of 3-year composite of CV events (HR: 0.81, p<0.001, Figure 3), statin discontinuation (HR: 0.81, p<0.001), and new-onset diabetes requiring medication (HR: 0.80, p=0.002) than high-intensity atorvastatin monotherapy7. The findings suggested that the combined therapy would result in more favorable outcomes and better drug compliance than high-intensity statin.
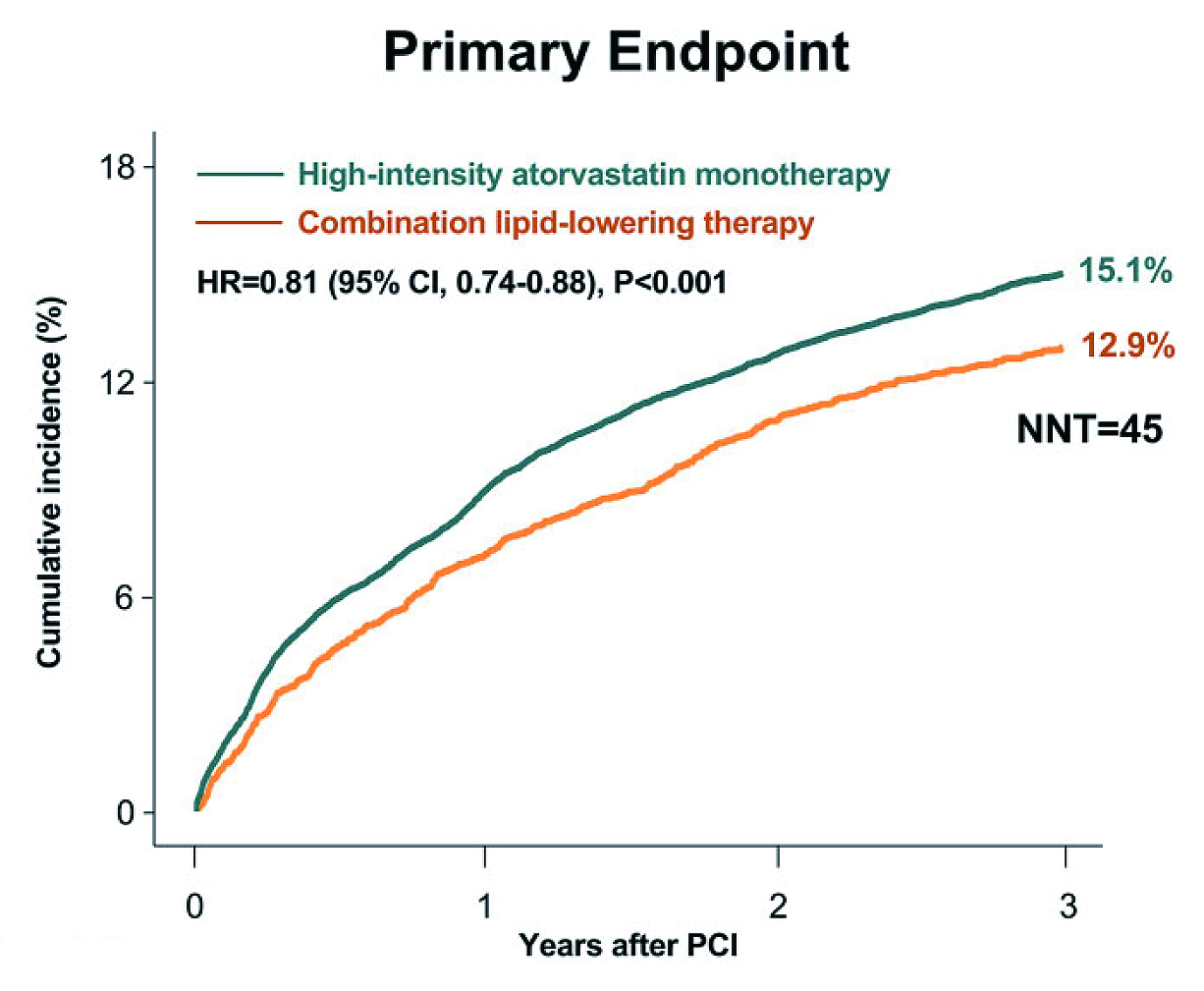
Figure 3: Time-to-event curves for composite of CV events7, NNT: number needed to treat
Prof. Lau commented that the results support the combined therapy as a first-line option. He further noted that the dosage of statin can be increased as appropriate. As the combined therapy of moderate-intensity atorvastatin and ezetimibe is now available as a single-pill regimen, the treatment is expected to improve patient adherence, particularly when polypharmacy is a concern.
Despite the established clinical benefits, the use of statins can be undermined in practice due to true or perceived intolerance. In this regard, the National Lipid Association (NLA) updated its working definition of statin intolerance in 2022, categorizing it as either complete or partial. Moreover, the NLA statement reasserts that a minimum of two statins should be trialed, at least one at the lowest approved daily dosage, to determine statin intolerance8.
Prof. Lau described that complete statin intolerance refers to cases in which statins are not tolerable at any lipid-lowering dose, whereas partial intolerance refers to the improved tolerance after a reduction in the maximally tolerated statin dose. While complete statin intolerance is practically rare, most patients can tolerate statins at certain dosages, and the side effects are dose dependent, which occurs more with high-intensity statins9.
For suspicious cases of statin intolerance, Prof. Lau advised ruling out any secondary causes of the reported symptoms first. “Some patients may have hypothyroidism or orthopedic problems, which lead to the symptoms. Some drug interactions may trigger the symptoms as well,” he noted. Then, a 2-week statin suspension can be trialed. If side effects persist, statin treatment can be resumed as the symptoms are unlikely to be statin related. Besides, if side effects are relieved with a reduced statin dosage, add-on therapy can be prescribed to maintain LDL-C lowering efficacy.
The 2025 Focused Update of ESC Guidelines redefines the CV risk categories and guides the treatment to achieve the specific LDL-C goals. Prof. Lau emphasized that LDL-C is a key modifiable risk factor for CV events. Thus, the lower LDL-C levels, the better. Particularly, early achievement of the LDL-C goal is needed for secondary prevention in high-risk patients. “Doublet or even triplet combined treatment may be needed for rapid LDL-C lowering in patients with extreme risk,” Prof. Lau commented.
As a final remark, Prof. Lau reminded that a high lipoprotein(a) [Lp(a)] level is an important risk modifier indicating increased CV risk, and a more aggressive treatment approach should be considered1. Practically, substantial LDL-C reduction is needed in most patients, and combined therapies are advisable. Regarding intolerance to high-intensity statins, moderate-intensity statins with add-on therapy can be considered. Last but not the least, Prof. Lau emphasized that lifestyle interventions are essential for all patients. “Red meats and fatty foods should be avoided, yet plenty of green vegetables and exercise are highly desirable,” he advised.
References
1. Mach F, Koskinas KC, Roeters van Lennep JE, et al. 2025 Focused Update of the 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Developed by the task force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Eur Heart J 2025; published online Aug 29. DOI:10.1093/EURHEARTJ/EHAF190. 2. Ouchi Y, Sasaki J, Arai H, et al. Ezetimibe Lipid-Lowering Trial on Prevention of Atherosclerotic Cardiovascular Disease in 75 or Older (EWTOPIA 75): A Randomized, Controlled Trial. Circulation 2019; 140: 992–1003. 3. Kytö V, Saraste A, Tornio A. Early statin use and cardiovascular outcomes after myocardial infarction: A population-based case-control study. Atherosclerosis 2022; 354: 8–14. 4. Her A-Y, Geol Choi B, Park S, et al. High-intensity statin on 3-year clinical outcomes in elderly acute myocardial infarction patients underwent percutaneous coronary intervention with drug-eluting stents. Scientific Reports 2025 15:1 2025; 15: 35325-. 5. Li S, Liu HH, Li JJ. Moderate-Intensity Statin Plus Ezetimibe: Time to Rethink it as an Optimal Initial Lipid-Lowering Strategy. Drugs 2025; 85: 51–65. 6. Mormone A, Tortorella G, Esposito F, et al. Advances in Pharmacological Approaches for Managing Hypercholesterolemia: A Comprehensive O. verview of Novel Treatments. Biomedicines 2024; 12: 432. 7. Lee SJ, Joo JH, Park S, et al. Combination therapy with moderate-intensity atorvastatin and ezetimibe vs. high-intensity atorvastatin monotherapy in patients treated with percutaneous coronary intervention in practice: assessing RACING generalizability. Eur Heart J Cardiovasc Pharmacother 2025; 10: 676–85. 8. NLA 2022 Definition of Statin Intolerance - American College of Cardiology. https://www.acc.org/latest-in-cardiology/articles/2022/08/08/12/27/nla-2022-definition-of-statin-intolerance (accessed Sept 26, 2025). 9. Cheeley MK, Clegg K, Lockridge C, Schubert TJ, Jones LK. Statin Intolerance: an Overview of US and International Guidance. Curr Atheroscler Rep 2023; 25: 517.
This material is for medical education with healthcare professionals only, supported by Organon HK Limited.
HK-ATO-110160 JAN/2026





