

Head of Department of Endocrinology-Diabetology,
Antwerp University Hospital & University of Antwerp, Belgium
Glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are the two incretin hormones, which stimulate pancreatic α and β cells and hence regulate insulin secretion with respect to blood glucose concentration. Accordingly, agonists stimulating receptors of GLP-1 and/or GIP and inhibitors of dipeptidyl peptidase-4 (DPP4), which prevents degradation of incretin hormones, have been developed to control type 2 diabetes (T2D)1. Remarkably, existing literature suggested that DPP4 inhibitors (DPP4is) have a favourable safety profile while effectively regulating glycaemic levels2. In a recent symposium titled “Seize Control of Type 2 Diabetes – Timing Matters”, Prof. Christophe De Block discussed the T2D management with incretin-based glucose-lowering medications.
Incretin and Its Roles in Countering T2D
Incretin effect is the enhanced insulin secretion with oral glucose than isoglycaemic intravenous (IV) glucose. Insulin secretion is regulated by incretin hormone (i.e., GLP-1 and GIP) based on blood glucose concentration. Meanwhile, the action of incretin hormones is tightly regulated, and the half-life of GLP-1 and GIP is shorter than 2 minutes due to the rapid action of DPP43.
Prof. De Block highlighted that, in T2D individuals, incretin effect is reduced, with a delayed insulin peak after oral glucose ingestion compared with healthy individuals (Figure 1A and 1B)1. The reduced incretin effect in T2D can be due to lack of or decrease in secretion of incretin hormone1. In this regard, there are two pharmacologic strategies countering T2D that use the incretin pathway. First, the action of GLP-1 can be enhanced by GLP-1 receptor agonists (GLP-1RAs), which bind and stimulate GLP-1 receptors and are resistant to DPP4 action. The second approach is to inhibit DPP4, which increases the level of active GLP-13.
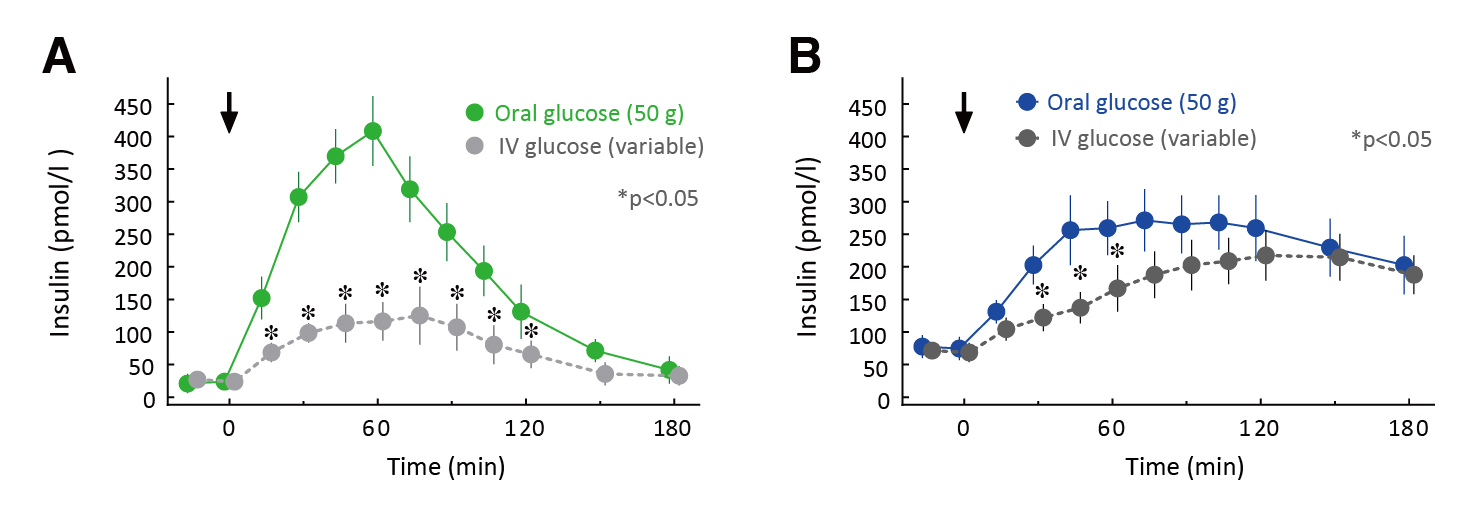
Figure 1. Incretin effect in A) healthy and B) T2D individuals1
The Pharmacology and Safety Profile of DPP4is
DPP4is only increase active GLP-1, resulting in elevated plasma GLP-1 levels at a concentration that minimises GLP-1-related side effects, such as nausea. Furthermore, the hypoglycaemia risk associated with DPP4is is minimal3.
Prof. De Block emphasised that a medication with higher selectivity is expected to minimise any potential off-target side effects4. For instance, sitagliptin has been reported to be highly selective for DPP4 with low inhibitory activity against DPP8 or 9 (Figure 2)5. Essentially, the enzymes DPP8 and 9 are very similar to DPP4, but inhibition of them produces severe toxicity in preclinical animal models5. “If I were to give a DPP4i, I would take into account the way it is metabolised and eliminated, its selectivity, and safety profile,” Prof. De Block advised.
-01.jpg)
Figure 2. In vitro selectivity of Sitagliptin5, DPP: dipeptidyl peptidase; FAP: fibroblast activation protein; PEP: prolyl endopeptidase; QPP: quiescent cell proline dipeptidase
Translating Pharmacology to Clinical Benefits
The GRADE study was a randomised open-labelled trial examining the relative effectiveness of four glucose-lowering medications (sitagliptin, glargine, glimepiride and liraglutide) when added to metformin in 5,047 patients with <10 years of T2D. In a secondary analysis of GRADE on insulin sensitivity and β-cell function (n=4,801), HbA1c was at the lowest level at year 1 in patients receiving liraglutide. However, the differences were eventually not significant at year 5 among the four treatment groups including sitagliptin (Figure 3A). Remarkably, the C-peptide index (CPI), which reflects β-cell function, was similar between liraglutide and sitagliptin and higher for both than for glargine and glimepiride (p<0.001, Figure 3B)6. Thus, the results suggested that insulin sensitivity remained stable after the first year while β-cell responses gradually declined over time. Interestingly, Stensen et al. (2022) reported that endogenous GIP improved β-cell function by 37±12% during DPP4 inhibition by sitagliptin7.
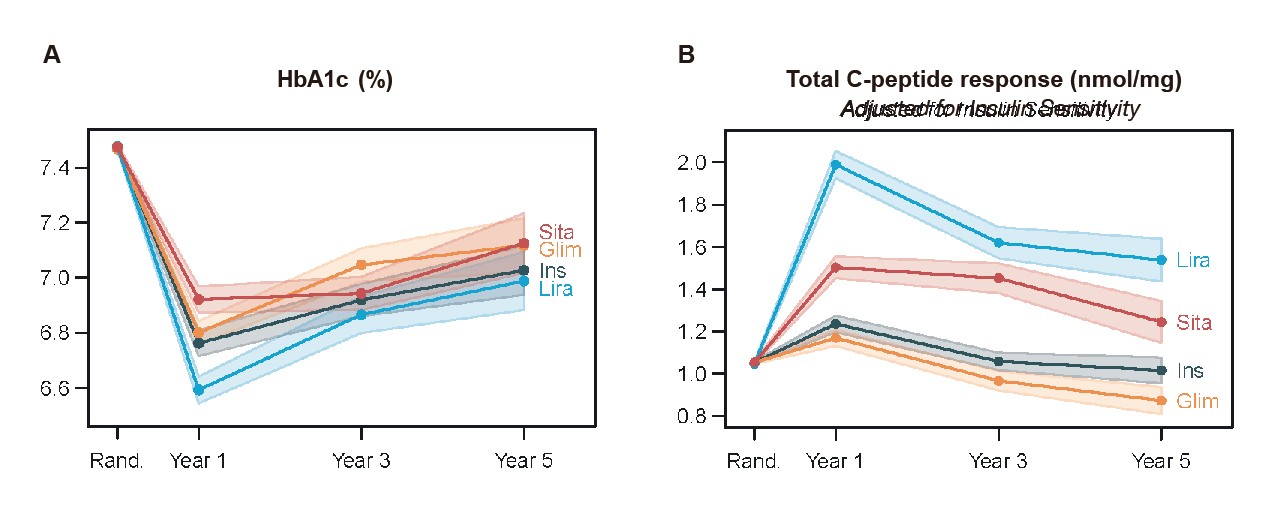
Figure 3. A) HbA1c and B) CPI upon glucose-lowering medications in GRADE study6, Rand: randomisation, Sita: sitagliptin, Glim: glimepiride, Ins: insulin glargine U-100, Lira: liraglutide
Further evaluating the clinical performance of sitagliptin, Prof. De Block highlighted its complementary effects with metformin, which yielded additive effects in reducing fasting and post-meal plasma glucose, augmenting GLP-1 secretion and β-cell function, decreasing plasma glucagon, and inhibiting fasting and post-meal endogenous glucose production than monotherapy of either treatment8.
Glycaemic Control – The Earlier, the Better
Prof. De Block advocated that early combination therapy with DPP4i and metformin can provide better glycaemic control. In the randomised, placebo-controlled CompoSIT-M study (2019) involving 458 patients who failed to achieve HbA1c goal on submaximal dose of metformin, patients were randomised to sitagliptin 100mg once daily or placebo, and all were to up-titrate metformin to 2000mg per day. The primary endpoint was the reduction from baseline in HbA1c. After 20 weeks of treatment, the least squares (LS) mean changes from baseline in HbA1c were -1.10% with sitagliptin versus -0.69% with placebo, with -0.41% of the between-group difference in LS mean changes from baseline (p <0.001) (Figure 4). Furthermore, patients in the sitagliptin group were more likely to achieve HbA1c <7.0% at week 20 than those in the placebo group in the overall population (relative risk [RR]: 1.7, p=0.002) and in the subgroup with a baseline HbA1c ≥8.5% (RR: 2.4, p=0.026)9.
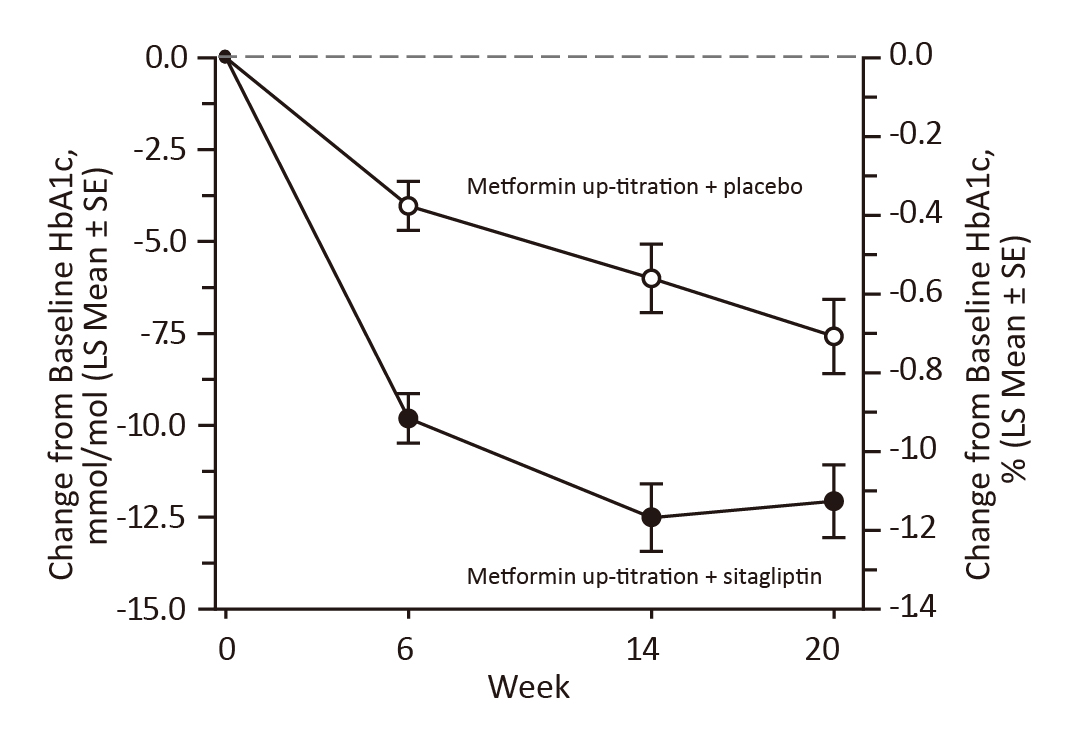
Figure 4. Glycaemic efficacy endpoints in CompoSIT-M study9, LS: least-squares; SE: standard error
Moreover, the randomised controlled trial by Reasner et al. (2011), which included 1,250 drug-naive patients with T2D, showed that initial therapy with the fixed-dose combination of sitagliptin and metformin yielded a significantly greater reduction in HbA1c from baseline at week 18, as compared to metformin monotherapy in T2D patients (-2.4% vs -1.8%, p<0.001). Notably, the incidences of diarrhoea and abdominal pain over 18 weeks were significantly lower for combination therapy versus metformin monotherapy10.
While hypoglycaemia is a clinical concern in managing diabetes, the previous trial by Nauck et al. (2007) involving 1,172 T2D patients with inadequate glycaemic control confirmed that sitagliptin was well-tolerated, with a significantly lower risk of hypoglycaemia relative to glipizide over 52 weeks (5% vs 32%, p<0.001)11.
Incretin and Its Roles in Countering T2D
The glycaemic impact of sitagliptin by race was assessed in the TECOS trial, which included 14,671 patients with T2D, cardiovascular disease and HbA1c of 6.5%-8.0%. Of these patients, 22.3% were from Asian countries, with >95% of self-identified East Asian patients being recruited from sites in the Asia-Pacific region. The patients were randomly assigned to receive sitagliptin or placebo. The results indicated that at 4 months, the mean placebo-adjusted HbA1c reduction was greater in East Asians (-0.60% versus ≤0.55% in other ethnic groups)12.
Furthermore, a pooled analysis of 20 placebo-controlled studies evaluating the clinical performance of sitagliptin specifically in East Asian populations by Conceicao et al. (2019) revealed that after 12 weeks of treatment, sitagliptin was associated with an overall reduction in HbA1c of -0.94% from a baseline HbA1c of 8.5%, which was significantly greater than that achieved with placebo (-0.22%, p <0.001)13. The clinical data thus highlighted in particular the efficacy of sitagliptin in East Asians.
Chronic kidney disease (CKD) is a common comorbidity among T2D patients. Accordingly, the safety of sitagliptin in patients with CKD was assessed in an analysis of the TECOS trial, which enrolled 14,671 participants, who were ≥50 years old with T2D, atherosclerotic cardiovascular disease, and HbA1c values of 6.5–8.0% on stable dose mono- or dual-combination therapy. The analysis compared participants assigned to sitagliptin versus those assigned to placebo with the CKD cohort. After 2.8 median years of follow-up, CKD patients who received sitagliptin (n=1,654) had similar rates of diabetic eye disease, diabetic neuropathy, renal failure, malignancy, bone fracture, pancreatitis, and severe hypoglycaemia as those who received placebo (n=1,642)14.
Case Sharing on Glycaemic Control
Case 1
To illustrate the clinical management of T2D, Prof De Block shared the cases of his patients. In the first case, a 57-year-old female schoolteacher who was diagnosed with T2D 4 years ago due to recurrent genitourinary tract infections (UTI). Her body-mass index (BMI) was 28 kg/m2, and HbA1c was 7.9%. Her renal function was normal. The patient was initially treated with metformin.
Prof De Block commented that the HbA1c was high, as the patient was still young. Thus, he decided to up-titrate the treatment. Given that sitagliptin effectively improves glycaemic control without causing weight gain, and increasing the risk of UTI and hypoglycaemia, Prof De Block prescribed an add-on sitagliptin for the patient. After 6 months, the patient well-tolerated the treatment and felt more energetic. The BMI was 27 kg/m2 and HbA1c was 7.1%. No impact on renal function was observed.
Case 2
The second patient was a 72-year-old male who was diagnosed with T2D 8 years ago. He was depressed due to his wife pass away and had lost 10 kg. His BMI was 21 kg/m2, and HbA1c was 8.1%. Mildly reduced renal function of 50 mL/min was documented. The patient was treated with metformin and diuretics.
Prof De Block opined that patient’s age should be considered, provided that the risk of reduced renal function and, hence, hypoglycaemia increase with age. Based on its established efficacy in older patients, Prof De Block prescribed an add-on sitagliptin. After 6 months, the patient reported to feel more energetic and less nocturia. His BMI (22 kg/m2) and HbA1c (7.4%) were well-maintained.
Timely Glycaemic Control for All
As incretins play a major role in the pathogenesis of T2D, selective inhibition of DPP4 allows glycaemic control with a preferable side effect profile. Existing evidence has demonstrated that DPP4is are effective both as monotherapy and in combination with metformin. Essentially, there is a minimal risk of hypoglycaemia associated with DPP4i. Remarkably, selective DPP4is, e.g. sitagliptin, have been proven effective in CKD patients without compromising safety profile and tolerability. In conclusion, Prof De Block opined that selective DPP4is can be used in a broad range of T2D patients, including East Asians. Earlier and intensive therapy is advisable to help patients reach HbA1c goals and improve outcomes, such as extending the time to treatment failure.
JANUVIA, JANUMET, JANUMET XR SSI
Indication: • JANUVIA (sitagliptin phosphate) is indicated as an adjunct to diet and exercise to improve glycemic control in patients with type 2 diabetes mellitus as monotherapy or in combination with metformin, or a PPARγ agonist, or a sulfonylurea, or insulin (with or without metformin), or a sulfonylurea and metformin, or a PPARγ agonist and metformin, when the current regimen, with diet and exercise does not provide adequate glycemic control or due to contraindications or intolerance. • JANUMET (sitagliptin phosphate/metformin HCl) and JANUMET XR (sitagliptin phosphate/metformin HCl extended release) are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus when treatment with both sitagliptin and metformin is appropriate. Contraindications: • JANUVIA, JANUMET and JANUMET XR are contraindicated in patients who are hypersensitive to any components of these products and should not be used in patients with pancreatitis, type 1 diabetes, acute or chronic metabolic acidosis (lactic acidosis) or for the treatment of diabetic ketoacidosis (with or without coma). • JANUMET and JANUMET XR are contraindicated in patients with severe renal impairment (eGFR below 30 mL/min/1.73 m2 or GFR < 30mL/min) or acute conditions with potential to alter renal function including dehydration, severe infection or shock. Lower JANUVIA dosages are recommended in patients with GFR < 45 mL/min, as well as in ESRD patients requiring hemodialysis or peritoneal dialysis. • JANUMET and JANUMET XR should be temporarily discontinued in patients undergoing radiologic studies involving intravascular administration of iodinated contrast materials or any surgical procedures (except minor procedures not associated with restricted intake of food and fluids). • Metformin should be discontinued prior to or at the time of the imaging procedure and not restarted until at least 48 hours after, provided that renal function has been re-evaluated and found to be stable. Precautions/ Warnings: (Post-marketing Experience/ General) • JANUVIA, JANUMET and JANUMET XR have been reported with serious hypersensitivity reactions include anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Acute pancreatitis with/without persistent and severe abdominal pain and bullous pemphigoid has been reported in patients taking sitagliptin. If any of the hypersensitivity reactions, pancreatitis with/ without symptoms, or any development of blisters, erosions or bullous pemphigoid is suspected, discontinue treatment, assess for other potential causes, and institute alternative treatment for diabetes. • For JANUVIA, Lower dosages are recommended in patients with GFR < 45 mL/min, as well as in ESRD patients requiring hemodialysis or peritoneal dialysis. • Hypoglycemia has been observed when sitagliptin and metformin (JANUVIA, JANUMET and JANUMET XR) were used in combination with insulin or a sulfonylurea or ethanol. Consider a lower dose of sulfonylurea or insulin to reduce the risk of sulfonylurea- or insulin-induced hypoglycemia. • If bullous pemphigoid is suspected, treatments should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment • Lactic acidosis may occur in diabetes mellitus, hepatic impairment, excessive alcohol intake, when there is a significant tissue hypoperfusion and hypoxemia, and due to metformin accumulation during treatment with JANUMET and JANUMET XR. It is characterized by elevated blood lactate levels (>5 mmol/L), decreased blood pH, electrolyte disturbances with an increased anion gap, and an increased lactate/pyruvate ratio. If metformin-associated lactic acidosis is suspected, discontinue treatment immediately. Adverse Events: • The most common adverse experience in sitagliptin monotherapy reported was nasopharyngitis. The most common (>5%) adverse reactions due to initiation of metformin therapy are diarrhea, nausea/vomiting, flatulence, abdominal discomfort, indigestion, asthenia, and headache. The most common adverse reactions in the combined extended-release metformin or placebo and PPARγ agonist were hypoglycemia, diarrhea and nausea. ◊ In the initial therapy of Combination Therapy with Sitagliptin and Metformin IR, patients may experience diarrhea, nausea, dyspepsia, flatulence, vomiting, headache and hypoglycemia. While in the add-on therapy, the incidences of pre-specified gastrointestinal adverse experiences in patients includes hypoglycemia, diarrhea, nausea, vomiting and abdominal pain. ◊ Sitagliptin in Combination with Metformin IR and a Sulfonylurea or Insulin: patients may experience hypoglycemia, constipation and headache. ◊ Sitagliptin in Combination with Metformin IR and a PPARγ Agonist: patients may experience headache, diarrhea, nausea, vomiting, hypoglycemia, upper respiratory tract infection, cough, fungal skin infection and peripheral edema. • JANUVIA was generally well tolerated in controlled clinical studies as both monotherapy and combination therapy.
Before prescribing, please consult the full prescribing information. Reference: S-HKPC-MK0431-T-052020, S-HKPC-MK0431A-T-052020, HKPC-MK0431A-XRT-122016-1
References
1. Nauck MA, Müller TD. Incretin hormones and type 2 diabetes. Diabetologia 2023;66:1780–95. 2. Yin R, Xu Y, Wang X, Yang L, Zhao D. Role of Dipeptidyl Peptidase 4 Inhibitors in Antidiabetic Treatment. Molecules 2022;27:3055. 3. Saini K, Sharma S, Khan Y. DPP-4 inhibitors for treating T2DM - hype or hope? an analysis based on the current literature. Front Mol Biosci 2023; 10. DOI:10.3389/FMOLB.2023.1130625. 4. Makrilakis K. The Role of DPP-4 Inhibitors in the Treatment Algorithm of Type 2 Diabetes Mellitus: When to Select, What to Expect. Int J Environ Res Public Health 2019; 16:2720. 5. Deacon CF. Dipeptidyl peptidase-4 inhibitors in the treatment of type 2 diabetes: a comparative review. Diabetes Obes Metab 2011;13(1):7-18. 6. Rasouli N, Younes N, Ghosh A, et al. Longitudinal Effects of Glucose-Lowering Medications on β-Cell Responses and Insulin Sensitivity in Type 2 Diabetes: The GRADE Randomized Clinical Trial. Diabetes Care 2024; 47: 580–8. 7. Stensen S, Gasbjerg LS, Rosenkilde MM, et al. Endogenous Glucose-Dependent Insulinotropic Polypeptide Contributes to Sitagliptin-Mediated Improvement in β-Cell Function in Patients With Type 2 Diabetes. Diabetes 2022; 71: 2209–21. 8. Solis-Herrera C, Triplitt C, De Jesús Garduno-Garcia J, Adams J, Defronzo RA, Cersosimo E. Mechanisms of glucose lowering of dipeptidyl peptidase-4 inhibitor sitagliptin when used alone or with metformin in type 2 diabetes: a double-tracer study. Diabetes Care 2013; 36: 2756–62. 9. Frias JP, Zimmer Z, Lam RLH, et al. Double-blind, randomized clinical trial assessing the efficacy and safety of early initiation of sitagliptin during metformin uptitration in the treatment of patients with type 2 diabetes: The CompoSIT-M study. Diabetes Obes Metab 2019; 21: 1128. 10. Reasner C, Olansky L, Seck TL, et al. The effect of initial therapy with the fixed-dose combination of sitagliptin and metformin compared with metformin monotherapy in patients with type 2 diabetes mellitus. Diabetes Obes Metab 2011; 13: 644–52. 11. Nauck MA, Meininger G, Sheng D, et al. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sulfonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: a randomized, double-blind, non-inferiority trial. Diabetes Obes Metab 2007; 9: 194–205. 12. Davis TME, Mulder H, Lokhnygina Y, et al. Effect of race on the glycaemic response to sitagliptin: Insights from the Trial Evaluating Cardiovascular Outcomes with Sitagliptin (TECOS). Diabetes Obes Metab 2018; 20: 1427–34. 13. Conceição JM, González CD, Engel SS, et al. 1188-P: Glycemic Efficacy of Sitagliptin in East Asian Populations: A Pooled Analysis. Diabetes 2019; 68. DOI:10.2337/DB19-1188-P. 14. Engel SS, Suryawanshi S, Stevens SR, et al. Safety of sitagliptin in patients with type 2 diabetes and chronic kidney disease: outcomes from TECOS. Diabetes Obes Metab 2017; 19: 1587–93.





