
BDS, MDS, PhD, FCDSHK, FHKAM
• Tam Wah-Ching Professor in Dental Science
• Professor, Chair of Dental Public Health
• Clinical Professor, The Faculty of Dentistry, the University of Hong Kong
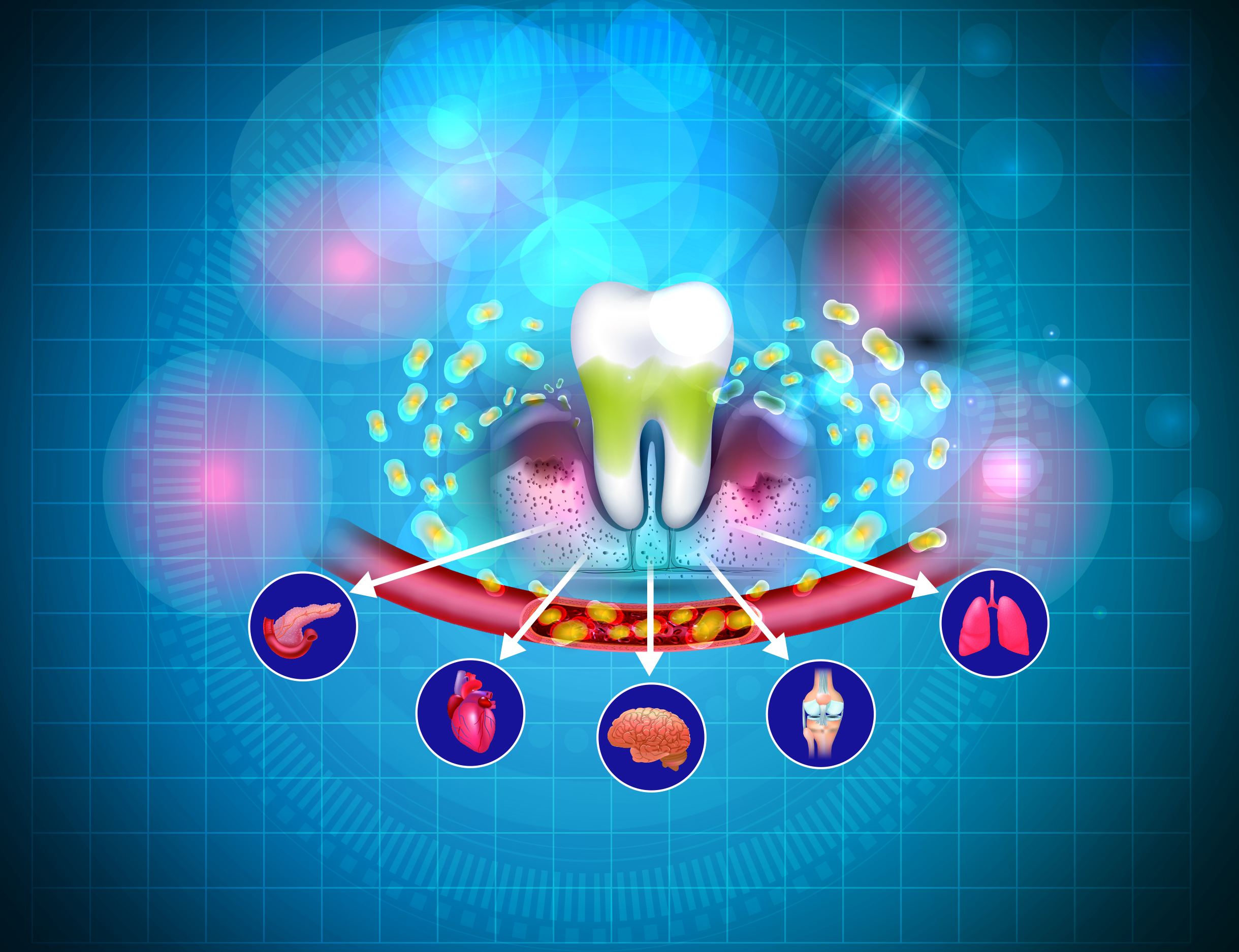
Periodontal disease (PD), also known as gum disease, is a chronic inflammation of the soft and hard tissues that support and anchor the teeth1. It affects up to 90% of the global population2. Gingivitis and periodontitis are the two main conditions within the PDs1. In Hong Kong, 97% of adults had bleeding gums with 40% having periodontal pockets3. Notably, periodontitis is the 6th most common human disease, globally4. Over the years, accumulating evidence has linked oral disease with many non-oral and systemic diseases, including neurodegenerative disorders5. Therefore, to understand the complexity of PDs, we have invited Professor Edward Chin Man Lo, a renowned dental expert to discuss the intricate link between oral health and systemic diseases, in addition to preventative measures taken to prevent PDs.
Background on Periodontal Diseases
PDs involve the periodontium, the supportive apparatus surrounding the tooth, and includes the gingival tissue, alveolar bone, cementum, and the periodontal ligament1. It is caused by the accumulation of biofilm as dental plaque on the tooth surface and along the gum line1. Superficial inflammation of the gums, known as gingivitis is a reversible condition that causes gum swelling and bleeding1. Contrarily, periodontitis occurs when the PD has progressed beyond gingivitis into a chronic, destructive, irreversible inflammatory disease state. Prof. Lo explained that bacteria including Porphyromonas gingivalis, Aggregatibacter actinomycetemcomitants, Treponema denticola, and Tannerella forsythia can colonise deep into tissues and the surrounding periodontium in patients with periodontitis, thereby, triggering a host response that ultimately leads to the destruction of periodontium1.
Patients with PD, particularly gingivitis often complain of experiencing red, swollen and bleeding gums that are painless. Patients with periodontitis may not exhibit any symptoms until the disease is in advanced stages. Prof. Lo reiterated that patients with periodontitis often present with symptoms of red, bleeding, as well as receding gums that are sometimes painful. He added that inflamed gums may start to pull away from the neck of the tooth which may then cause gaps to form between teeth and the gums, also known as periodontal pockets. As the disease advances, teeth position may shift and they may start to wobble or patient may complain of pain when chewing6. Therefore, it is paramount to accurately detect the presence of the disease during its early stages. Notably, the diagnosis of PD is based on the clinical and radiographic measures. “Clinical assessment of periodontal soft tissues on periodontal probing enables clinician to detect irregularities, tooth mobility, and migration in patients with periodontitis,” according to Prof. Lo.
Unravelling the Link Between Periodontal Diseases and Systemic Diseases
Over the last few years, accumulating evidence has linked oral diseases with many non-oral and systemic diseases and conditions, including cardiovascular disease (CVD), type 2 diabetes (T2D), obesity, rheumatoid arthritis (RA), osteoporosis, respiratory infections, inflammatory bowel disease (IBD), Alzheimer's disease (AD), non-alcoholic fatty liver disease (NAFLD),, chronic kidney disease (CKD) and certain cancers7. An 18-year retrospective cohort study by Zhao et al., (2019) evaluated whether poor periodontal conditions may increase the risk for onset of systemic comorbidities.8 The study included data from 488 dental attendees who were randomly selected from 17,400 dental hospital attendee from the period 1996 to 1998. These attendees were free of diabetes, CVD, chronic obstructive pulmonary disease (COPD), cancer, stroke, cognitive impairment, hypertension and dyslipidaemia in the Clinical Management System. The results demonstrated that participants with worse periodontal status on their first registration had significantly higher number of the 8 comorbidities/mortality during the follow-ups than their counterparts (p<0.05). Moreover, those with onset of more comorbidities showed worse periodontal conditions, according to four parameters employed (Community Periodontal Index, Bone Loss, Bone loss/age and Number of remaining teeth) (p<0.05)8. Similarly, a cross-sectional study by Zhao et al., (2022) suggested that patients with worse PD status showed the worse medical diagnostic test results than their counterparts9; thereby, a coordinated effort by both oral and medical health professionals in enhancing health awareness is important.
Further links between PDs and systemic diseases were reported in a systemic review and meta-analysis by Serni et al., (2023) evaluating the association of periodontitis at different stages of CKD. The study found that CKD was associated with a higher risk of periodontitis (Mantel-Haenszel-Odds ratio [MH-OR] = 2.36 bring on same line as above, [95% confidence interval (CI): 1.25, 4.44]; p = 0.008), higher mean clinical attachment level (CAL) (weighted mean different [WMD] = 0.41 mm [95% CI: 0.22, 0.60]; p<0.0001) and mean periodontal probing depth (PPD) (WMD = 0.25 mm [95% CI: 0.03, 0.47]; p=0.02) compared to healthy individuals. Furthermore, those with higher risk of PD often had severe CKD (stages 4-5 versus stage 2-3) (p=0.03). The study concluded that there is an association between periodontitis and CKD and periodontitis should be frequently considered as a comorbidity in CKD10. The reason behind this relationship stems from the fact that these two conditions share several risk factors, in addition to patients with CKD having an increased gingival crevicular fluid levels of urea, thereby generating an alkaline pH environment, promoting the growth of periodontal pathogens such as P. gingivalis7. These findings are not isolated to CKD only since dental plaque biofilm has been proposed to act as a reservoir for respiratory infections considering oral microbes had been isolated from secretions of pneumonia patients7. The negative effects of periodontitis has also shown to exert adverse pregnancy outcome in women since periodontitis causes elevated levels of pro-inflammatory molecules in the maternal circulation that may inadvertently induce the production of inflammatory cytokines in foetal-placenta unit, leading to miscarriages and stillbirths7.
Critical Link Between Periodontal Disease and Cardiometabolic Disorder
Recent epidemiological data has suggested a bidirectional association between periodontitis and cardiometabolic disorders as translocation of bacterial lipopolysaccharide (LPS) into the blood circulation may cause endotoxemia, which may then increase the patient risk of developing cardiometabolic disorder7. These findings were substantiated in a 9-year-follow-up study by Morita et al., (2016) that examined the relationship between the duration of periodontitis and cardiometabolic risk factors, including hypertension, hyperglycaemia, dyslipidaemia and obesity11. The study included 572 adult industrial workers (417 males and 155 females, mean age 37.4 years) who had undergone annual medical and dental health examinations from 2003 to 2012. Remarkably, the results showed that the OR for the presence of ≥ 1 cardiometabolic risk factor in patients with cumulative duration of periodontal pocket for ≥ 6 years was significantly higher than participants without pockets. The ORs for the onset of obesity, hypertension, dyslipidaemia, and hyperglycaemia were higher in participants with a cumulative duration of periodontal pockets for ≥ 6 years than participants without pockets or in participants with a cumulative duration of periodontal pockets for ≤ 5 years. This suggests that individuals with untreated periodontitis are at greater risk of developing cardiometabolic disorders.
PD is now also being considered as the 6th major complication of diabetes mellitus (DM) since DM may worsen the virulence and invasiveness of pathogenic oral microbial flora, aggravating local inflammation and infection in those with PD. On the other hand, the chemical and immunological mediators released into the circulation secondary to periodontal inflammation may worsen insulin resistance among diabetics. Remarkably, the incidence of macroalbuminuria and end-stage renal disease are twofold and threefold higher in diabetics with severe periodontitis compared to diabetics without severe periodontitis12. The intimate relationship between DM and periodontitis was corroborated in a systematic review by Alwithanani et al., (2023) evaluating how poorly controlled diabetes affected the progression of periodontitis. The review included 13 studies with 49,262 participants of which 3,197 were diabetics. Interestingly, the results showed that diabetes may augment the likelihood of developing or progressing into periodontitis by up to 86% (risk ratio [RR] 1.86; 95% CI: 1.3-2.8)13. Considering the critical link between DM and periodontitis, referral to dental health professionals for treatment may serve as an important novel tool for medical professionals in preventing and managing DM14.
Bridging the Gap Between Periodontal Disease and Dementia
Clinical and microbial markers of PD have been associated with the incidence and mortality of Alzheimer's disease (AD). In fact, periodontitis has been considered to be one of the probable risk factor for AD15 since periodontitis may aggravate AD pathology by increasing the systemic inflammation7. This relationship was explored in systematic review by Borsa et al., (2021) that reported periodontitis at baseline was associated with a six-fold increase in the rate of cognitive decline over a 6-month follow-up period (the Alzheimer’s Disease Assessment Scale–Cognitive Subscale [ADAS-Cog] mean change = 2.9 ± 6.6)16.
Chronic inflammatory response in periodontitis may potentially be a risk factor for dementia. This was studied further by Chen et al., (2023) who evaluated the association between periodontal treatment and dementia, in addition to the effect of regular scaling treatment had on the risk of dementia in 18,930 patients with a dementia-related diagnosis from the Taiwan National Health Insurance Research Database17. Remarkably, the results showed that scaling treatment rates were lower among the dementia cohorts than non-dementia cohorts after 1 and 3 years. More astonishingly, patients who received periodontal emergency treatment within 3 years exhibited a significantly increased risk of dementia (OR 1.06, 95% CI: 1.00-1.12). Furthermore, patients with periodontitis who had not received any scaling treatment within 3 years were at a higher risk of developing dementia compared to patients without periodontitis (OR 1.22; 95% CI: 1.10-1.35)17 (Figure 1). The findings demonstrated that periodontitis and dementia share a close relationship, and periodontitis is considered a risk factor for dementia. Additionally, these findings also highlighted the importance of regular and routine scaling treatment17, Prof. Lo commented.
-01.jpg)
Figure 1. Ratios of dementia among different periodontal treatment. Group 1 (had scaling mouth but no periodontal treatment), group 2 (had no scaling mouth, and no periodontal treatment), group 3 (had both scaling mouth and periodontal treatment), and group 4 (had no scaling but had PD)17. PD, periodontal disease.
Prevention is Always Better Than Cure
Considering periodontitis is a chronic multifactorial disease characterised by the accumulation of dental biofilm and progressive destruction of the teeth-supporting apparatus, it is paramount for a comprehensive periodontal evaluation18. Treatment of periodontitis is aimed at preventing the disease progression with intention to reduce the risk of tooth loss, minimise symptoms and perception of the disease and possibly restore lost periodontal tissues19. Prof. Lo added that good oral hygiene is the cornerstone of PD prevention through simple measures such as tooth brushing as per the American Dental Association (ADA) guidelines20, flossing, and using mouth wash, in addition to visually inspecting and recognising the individual modifiable risk factors21. Although regular toothpaste formulations often contain a combination of fluorides and detergents to enhance the enamel strength, efficacy of brushing, and thereby prevent diseases, brushing efficacy can be enhanced further by the addition of antibacterial substances in toothpaste which can aid oral biofilm removal further. Prof. Lo commented that chlorohexidine digluconate (CHX) addition to mouthrinse is considered a “gold standard” for oral cavity antiseptic treatment, especially before and after dental surgical procedures to prevent oral ulcers and manage intensive periodontal diseases. Other antimicrobial agents such as zinc (Zn) provides antibacterial properties and anti-matrix metalloproteinase activity when used as a coating metal, promoting an inhibitory effect on gram-negative anaerobes. In addition, it also exerts an inhibitory effect on oral malodour by directly binding with the gaseous dihydrogen sulphide (H2S) and sulphur producing oral bacteria22. Toothpaste with stannous fluoride has been shown to have further positive effects on reducing dental calculus build-up, dental plaque, gingivitis, stain, halitosis, recalcification of enamel, in addition to reduce biofilm formation as per recent studies23,24.
Aside from stannous fluoride, sodium bicarbonate is an additional unique compound added to toothpastes to aid plaque removal. It is hypothesised to physically displace plaque from the tooth surface and/or interfere with the adhesion characteristics of the biofilm leading to a reduction of biofilm structural integrity. These properties of sodium bicarbonate were assessed in a pooled analysis of 6 randomised controlled trials (RCTs) by Parkinson et al., (2022) on antigingivitis and anti-plaque treatment effect of a toothpaste containing 67% weight per weight (w/w) sodium bicarbonate versus non-sodium bicarbonate (regular) toothpaste, at the individual tooth site, tooth region and whole mouth (overall) level. The results demonstrated that sodium bicarbonate toothpaste effectively removed plaque from all tooth sites, and resulted in a clinically significant improvement in measures of gingival health, overall and for all the tooth regions investigated, compared with a non-sodium bicarbonate (regular) toothpaste following 24 weeks twice daily use25. Other compounds recently added to toothpaste include isopropyl-3-methylphenol (IPMP or o-Cymen-5-ol) and zinc, which has also demonstrated bactericidal action against microorganisms such as Porphyromonas gingivalis, Fuscobacterium nucleatum, Streptococcus mutans26. A study by Kakar et al., (2011) assessed the ability of 0.1 w/w o-cymen-5-ol/0.6 w/w zinc chloride dentifrice to maintain gingival health compared to a sodium fluoride control dentifrice in 224 participants. The results demonstrated that 0.1% w/w o-cymen-5-o1/0.6% w/w zinc chloride dentifrice over a 12 week period provides a statistically significant benefit in maintaining gingival health compared to a sodium fluoride control dentifrice27.
Hyaluronic acid (HA) is another naturally occurring compound shown to help maintain the structural, homeostatic integrity of tissues, act as anti-inflammatory, anti-oedematous, as well as anti-bacterial compound for the treatment of PD (Figure 2)28. In fact, recent study data has shown HA to have regenerative function in periodontal tissues when used as the treatment of PD28. Furthermore, HA plays a pivotal role in the postoperative care of patients undergone dental procedure, as well as patients with chronic inflammatory gingival and oral ulcers29. The unique properties of HA were reported in a study by Mehta et al., (2022) where HA exhibited better results in complete root coverage and mean root coverage when compared to the control group in periodontal surgery. Remarkably, gingival recession reduction, clinical attachment level, keratinised tissue gain was significantly increased compared to the control group30. HA is believed to bind to the CD44 receptor that effects the coating of cell membrane. This protective layer of hyaluronan on cell surface is then able to mask the cell death receptors and consequently prevent cells from reaching apoptosis31.
-01.jpg)
Figure 2. Properties of hyaluronic acid28
Common Substances in Toothpastes for Achieving Optimal Periodontal Health
|
Toothpaste Compounds |
Mechanism of action |
|
Zinc ion (Zn2+) |
The antibacterial activity of Zn2+ is related to its ability to bind gaseous hydrogen sulphide (H2S) and inhibit H2S volatilisation. However, the effect on growth of oral bacteria was strain-dependent (F.nucleatum being the most sensitive). Zn2+ may have an inhibitory effect on oral malodor by directly binding to gaseous H2S and a volatile sulphur compound-producing oral bacteria suppression22. |
|
Stannous fluoride (SnF2) |
SnF2 reduces the quantity of dental plaque and modulates the metabolic activity of dental plaque. In addition it has a strong affinity to directly bind bacterial endotoxins, such as lipopolysaccharides (LPS), causing suppression of Toll-like receptors, which prevents activation of pathways that cause gingival inflammation32. |
|
Sodium bicarbonate (NaHCO3) |
NaHCO3 acts as cleansing agent, dissolving mucus and loosening debris. Some postulate that it may work against the plaque biofilm by disrupting the exopolysaccharide matrix structure. In addition, it can disrupt mature dental plaque growth in vitro, removing substantial amount of mature biofilm33. |
|
Isopropyl methylphenol (IPMP) |
IPMP is a typical preservative and bactericidal constituent with phenolic compound believed to exhibit antimicrobial activity by damaging cell membrane, and changing the membrane and function34. |
|
Hyaluronic acid (HA) |
HA is a mucopolysaccharide present in many areas of the body and has a wide range of anti-inflammatory effects, promote tissue regeneration and repair35. It is a ligand for receptor CD44 and enhances the mineralisation of dental pulp cells mediated via CD4436. Exogenous use of HA binds to CD44 in epidermal or fibroblast cells, causing a signal transduction mediated by the CD44 to promote wound healing37. |
Multidisciplinary Approach to Periodontal Disease
The provision of preventive oral healthcare advice has been traditionally provided by the dental health professionals; however, over the recent years, a links between oral health and a number of systemic diseases has been established7. Considering PDs and systemic diseases can virtually be painless, especially at early stages as demonstrated in a study by Zhao et al., (2022) where individuals in Hong Kong self-perceived as healthy, but in reality, 98% had undiagnosed systemic diseases/disorder, while 96% had periodontitis9. A multidisciplinary approach is often required since the general practitioners (GPs)38 and community pharmacists can provide basic oral health advices and interventions that may help change oral health behaviours among patients whom oral health has not been a priority39. Prof. Lo emphasised that community pharmacists are in a unique position to disseminate information on oral health since they are in the frontline of primary source of information for many patients.
In addition, pharmacists, GPs, nurses may help identify, assess and refer patients with PD when necessary. Although they cannot substitute the role of a dental health professional, they can still distribute information related to preventative measures, and the importance of regular dental check-ups40. Since patients with dental problems often consult their GP or community pharmacists due to various reasons, including management of comorbidities and concerns of the financial cost of dental treatment. Therefore, taking a multidisciplinary approach through collaborative services between non-dental health professionals and dentists may help provide a high-quality health services to patients, in addition to reduce the disease burden of PDs41. In summary, there are well-established and scientifically proven approaches (tooth and implant cleansing, topical chemotherapeutics, reduction in risk factors, such as tobacco smoking) to reducing the risk of PD, and its intricate link with the systemic diseases42. Furthermore, recent study data has shown that severe PD is associated with a 25-90% increase in risk for CVD43. Therefore, dental health, and non-dental health professionals are in a unique position to promote oral and systemic health through collaborative effort and maximise the benefit of oral health prevention.
References
1. WHO. Global oral health status report. https://www.who.int/team/noncommunicable-diseases/global-status-report-on-oral-health-2022 2. Gasner NS, et al. StatPearls Publishing LLC.; 2024 3. Gao SS, et al. Oral Health Care in Hong Kong. Healthcare 2018; 6(2):45. 4. Sanz M, et al. J Clin Periodontol 2020; 47(3): 268-88. 5. Bui FQ, et al. Biomedical Journal 2019; 42(1): 27-35. 6. (IQWiG).Gingivitis and periodontitis. 2023. https://www.ncbi.nlm.nih.gov/books/NBK279593/ 7. Hajishengallis G. et al. Periodontol 2000 2022; 89(1): 9-18. 8. Zhao D, et al. J Periodontol 2019; 90(3): 225-33. 9. Zhao D, et al. Journal of Periodontal Research 2022; 57(3): 632-43. 10. Serni L, et al. Oral Dis 2023; 29(1): 40-50. 11. Morita T, et al. Metab Syndr Relat Disord 2016; 14(10): 475-82. 12. Preshaw PM, et al. Diabetologia 2012; 55(1): 21-31. 13. Alwithanani N. J Pharm Bioallied Sci 2023; 15(Suppl 1): S54-S63. 14. Borgnakke WS. Diabetes Research and Clinical Practice 2019; 157: 107839. 15. Abbayya K, et al. N Am J Med Sci 2015; 7(6): 241-6. 16. Borsa L, et al. Int J Environ Res Public Health 2021; 18(17): 9312. 17. Chen H-L, et al. BMC Oral Health 2023; 23(1): 969. 18. Kwon T, et al. Int Dent J 2021; 71(6): 462-76. 19. Graziani F, et al. Periodontol 2000 2017; 75(1): 152-88. 20. Van Leeuwen MPC, et al. Int J Dent Hyg 2019; 17(1): 77-84. 21. Janakiram C, et al. Periodontol 2000 2020; 84(1): 202-14. 22. Caruso S, et al. Materials 2024; 17(4): 800. 23. Fiorillo L, et al. Biomimetics 2020; 5(3): 41. 24. Johannsen A, et al. Heliyon 2019; 5(12): e02850. 25. Parkinson CR, et al. Int J Dent Hyg 2023; 21(1): 106-15. 26. Pizzey RL, et al. Int Dent J 2011; 61(Suppl 3): 33-40. 27. Kakar A, et al. International Dental Journal 2011; 61: 13-20. 28. Dahiya P, et al. N Am J Med Sci 2013; 5(5): 309-15. 29. Casale M, et al. Int J Immunopathol Pharmacol 2016; 29(4): 572-82. 30. Mehta V, et al. Int J Environ Res Public Health 2022; 19(21): 14330. 31. Litwiniuk M, et al. Wounds 2016; 28(3): 78-88. 32. Xie S, et al. Front Oral Health 2024; 5: 1427008. 33. Pratten J, et al. Int J Dent Hyg 2016; 14(3): 209-14. 34. Ohara H, et al. PLoS One 2023; 18(9): e0283295. 35. Miglani A, et al. Cureus 2023; 15(10): e46349. 36. Chen KL, et al. J Endod 2016; 42(5): 711-6. 37. Kawano Y, et al. Pharmaceuticals 2021; 14(4): 301. 38. Tadin A, et al. Dent J 2023; 11(11): 259. 39. Sturrock A, et al. BDJ Open 2020; 6: 13. 40. Bawazir OA. J Int Oral Health 2014; 6(6): 10-3. 41. Rezvaninejad R, et al. BMC Oral Health 2024; 24(1): 1130. 42. Scannapieco FA, et al. Periodontol 2000 2020; 84(1): 9-13. 43. Kim J, et al. Odontology 2006; 94(1): 10-21.





