

Department of Hematology and Rheumatology,
Kindai University, Faculty of Medicine,
Japan

Department of Hematology, Nephrology, and Rheumatology,
Akita University Graduate School of Medicine and Faculty of Medicine, Japan

Chief of the Division of Hematology, Oncology, and Bone Marrow Transplantation,
Department of Medicine,
School of Clinical Medicine,
The University of Hong Kong (HKU), Hong Kong

Hematology Centre, Department of Hematology, Uijeongbu Eulji Medical Center,
Leukemia Omics Research Institute,
Eulji University, South Korea

Department of Hematology, Nanfang Hospital,
Southern Medical University, China

Department of Hematology,
The First Affiliated Hospital of
China Medical University,
China
Ponatinib is a third-generation tyrosine kinase inhibitor (TKI) approved as a treatment for chronic myeloid leukemia (CML) patients with resistance or intolerance (r/i) to prior TKIs and patients with relapsed and refractory Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL). Notably, recent studies have demonstrated favorable safety and efficacy of ponatinib in Japanese patients. Based on these findings, an advisory board meeting was conducted on the 20th April 2024, that invited renowned hematology experts from Asia (China, Hong Kong, Japan and South Korea) to discuss the current status and future challenges in Ph+ ALL and CML treatment.
The development of TKIs for the treatment of CML or Ph+ ALL has led to the introduction of a selective targeted therapy aimed to suppress the growth of leukemic cells1. Ponatinib, a third-generation TKI, was approved in Japan by the Ministry of Health, Labour and Welfare in September 2016 as a treatment for CML patients with r/i to prior TKIs and patients with relapsed and refractory Ph+ ALL1. The post- marketing all-case surveillance on the safety and efficacy of ponatinib in clinical practice, focusing on arterial occlusive events (AOEs) was presented in a study that included data from 724 Japanese CML patients with r/i to prior kinase inhibitors and patients with relapsed/refractory (r/r) Ph+ ALL1. Remarkably, among patients in chronic phase of CML (CML-CP), 11.9% carried the T315I mutation. In addition, 25.4% of patients with CML-CP and 27.4% of patients with Ph+ ALL were hypertensive. Notably, adverse drug reactions (ADRs) were reported in 58.29% of patients with decreased platelet count, hypertension, rash, decreased neutrophil count and abnormal hepatic function being the most common ADRs1. Surprisingly, the incidence of AOEs was 6.49% among the overall population, 6.22% among patients with CML-CP and 5.90% among patients with Ph+ ALL (Figure 1)1.
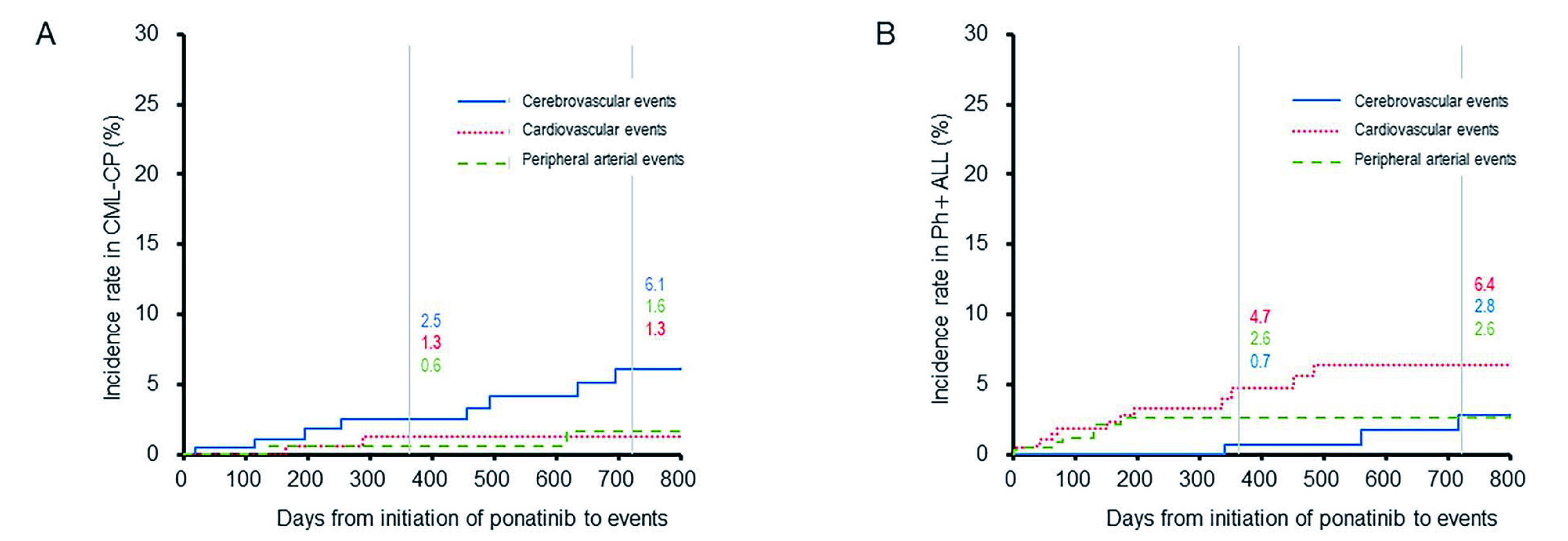
Figure 1. Cumulative incidence rates of arterial occlusive events in patients with CML-CP and Ph+ ALL. The cumulative incidence rates of cerebrovascular (solid line), cardiovascular (dotted line), and peripheral arterial events (dashed line) are shown in patients with CML-CP (A) and Ph+ ALL (B). Incidence rates are labeled at years 1 and 21. CML=chronic myeloid leukemia; CP=chronic-phase; Ph+ ALL=Philadelphia chromosome-positive acute lymphoblastic leukemia.
Among patients with CML-CP who had not achieved major molecular response (MMR) prior to treatment with ponatinib, the cumulative MMR rate at weeks 12, 24, 52 and 104 was 22.8%, 36.9%, 58.8%, and 67.2% (Figure 2), respectively1. Furthermore, in 19 patients with T315I mutation, the cumulative MMR rate at 12, 24, 52, and 104 weeks were 42.1%, 47.4%, 75.3%, and 75.3% respectively. In summary, the study demonstrated a generally favorable safety and efficacy of ponatinib in Japanese patients.
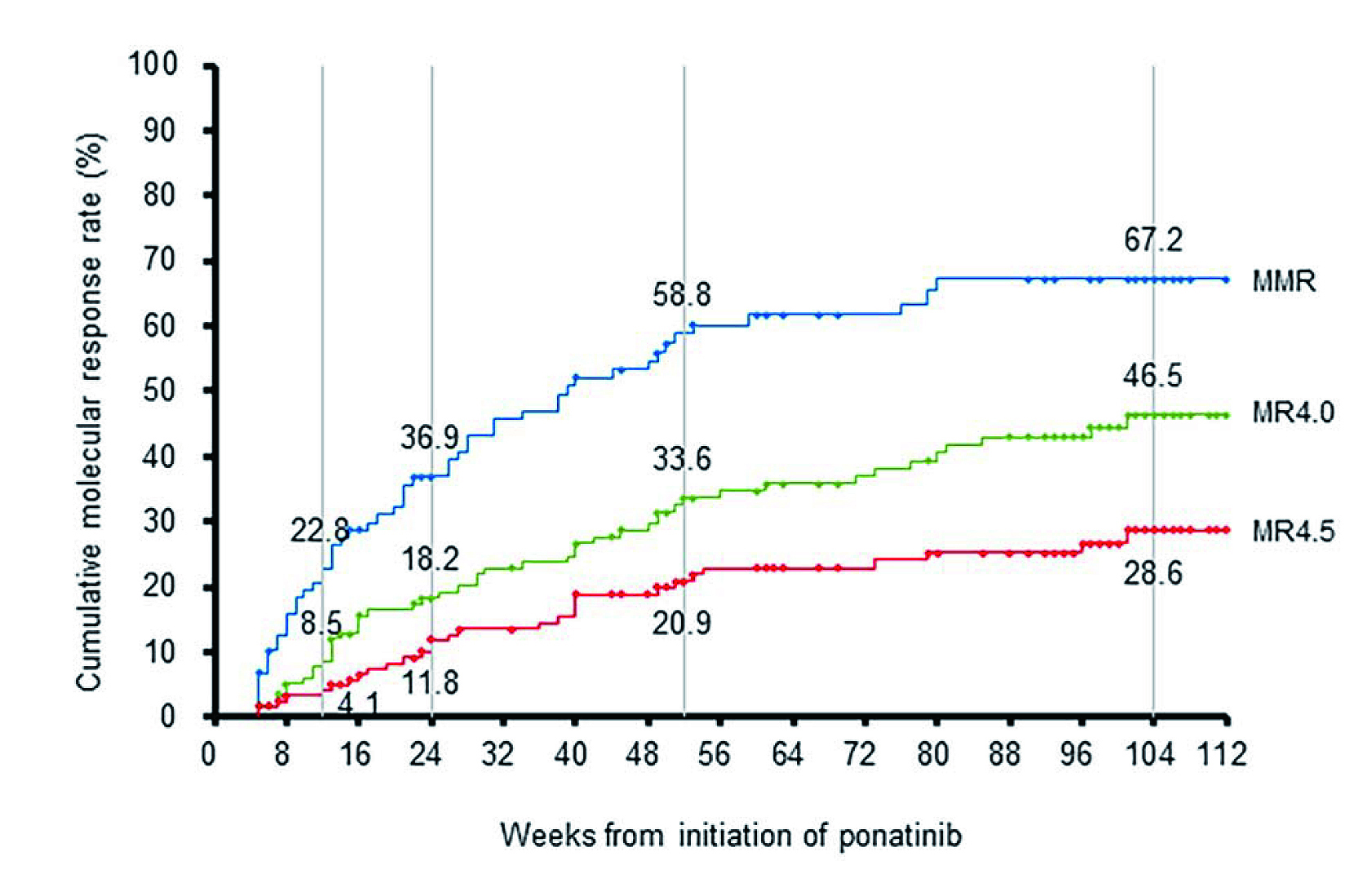 Figure 2. Cumulative molecular response rates in patients with CML-CP who did not achieve MMR to prior treatment. The cumulative MMR rate, MR4.0 and MR4.5 rates in CML-CP patients who did not achieve MMR before treatment with ponatinib are shown. Response rates are labeled at weeks 12, 24, 52, and 1041. CML= chronic myeloid leukemia; CP=chronic-phase; MMR= major molecular response (BCR::ABL1 transcripts international scale [IS] ≤ 0.1%); MR4.0= molecular response with a 4.0-log reduction (BCR::ABL1 transcripts [IS] ≤ 0.01%); MR4.5= molecular response with a 4.5-log reduction (BCR::ABL1 transcripts [IS] ≤ 0.0032%).
Figure 2. Cumulative molecular response rates in patients with CML-CP who did not achieve MMR to prior treatment. The cumulative MMR rate, MR4.0 and MR4.5 rates in CML-CP patients who did not achieve MMR before treatment with ponatinib are shown. Response rates are labeled at weeks 12, 24, 52, and 1041. CML= chronic myeloid leukemia; CP=chronic-phase; MMR= major molecular response (BCR::ABL1 transcripts international scale [IS] ≤ 0.1%); MR4.0= molecular response with a 4.0-log reduction (BCR::ABL1 transcripts [IS] ≤ 0.01%); MR4.5= molecular response with a 4.5-log reduction (BCR::ABL1 transcripts [IS] ≤ 0.0032%).
Currently, there are multiple TKIs available for the Ph+ ALL, which includes imatinib (a first-generation TKI), dasatinib, nilotinib (second-generation TKIs) and ponatinib (third-generation TKI)2. But which TKIs are recommended for the newly diagnosed Ph+ ALL patients? Prof. Kim, a prominent hematology expert from Uijeongbu Eulji Medical Centre, Leukemia Omics Research Institute in South Korea explained that imatinib is the only available TKI for newly diagnosed Ph+ ALL patients and it is often administered at a high dose concurrently with chemotherapy3 in South Korea. He added that if patients develop resistance to imatinib, or are unable to tolerate imatinib-based therapy, dasatinib4 and ponatinib are initiated5. Similarly, Prof. Yan, a hematologist from The First Affiliated Hospital of China Medical University outlined the local practice in China as she highlighted the fact that even though the health insurance in China covers both first- and second-generation TKIs, patients often prefer the second-generation TKIs as the first-line treatment due to their higher efficacy compared to the first-generation TKIs6. She also commented that even though the ponatinib is efficacious7, it is currently not available in China. Prof. Liu, a hematology expert from the Nanfang Hospital, Southern Medical University in China explained that ponatinib is also associated with an increased risk of hypertension and thromboembolism8; however, she emphasized that these side effects are milder in Chinese patients compared to the European and American patients.
Prof. Kwong, chief of the Division of Hematology, Oncology and Bone Marrow Transplantation at the Department of Medicine, HKU, shared the local treatment landscape of newly diagnosed Ph+ ALL patients in Hong Kong. He explained that imatinib plus chemotherapy is initially used for newly diagnosed Ph+ ALL patients, and if they develop resistance to imatinib, a second-generation TKI such as dasatinib or nilotinib is added. Here he stressed that the goal is for Ph+ ALL patients to receive allogeneic hematopoietic stem cell transplantation (allo-HSCT). However, some patients may receive adolescents and young adults (AYA) type treatment plus ponatinib, and these patients may become minimal residual disease (MRD) negative; thus, not requiring the need for allo-HSCT, according to Prof. Kwong. He elaborated that the AYA type treatment consists of an induction therapy followed by consolidation, an interim maintenance, delayed intensification and long-term maintenance therapy9. He then went on to explain the 3 approaches that can be taken to treat newly diagnosed Ph+ ALL patients.
The first approach is to offer patients the first- or second-generation TKI plus chemotherapy, which is then followed by allo-HSCT, regardless of their MRD negativity. The second approach is to offer patients the AYA type treatment plus ponatinib and if they become MRD negative, they are exempted from allo-HSCT. The final approach is for patients who are unable to tolerate chemotherapy or are too old or have other comorbidities, where blinatumomab plus ponatinib is offered and if they become MRD negative, they are not recommended for allo-HSCT. Here, Prof. Kwong stressed that if newly diagnosed Ph+ ALL patients are treated with the AYA type treatment involving a combination of either nilotinib or dasatinib, they would still be recommended for allo-HSCT since there is an uncertainty whether the same depth of remission can be obtained with these agents compared to ponatinib. Prof. Matsumura, the chair of the advisory board meeting, and a prominent hematology expert from the Department of Hematology and Rheumatology at Kindai University in Japan clarified that ponatinib can prevent the emergence of T315I mutation10, hence, there is an uncertainty whether other agents highlighted by Prof. Kwong can achieve that.
Approximately, half of the elderly patients with ALL have the Ph chromosome, making it the most common cytogenetic abnormality in this age group11. Prof. Matsumura explained that addition of very mild steroids to TKI treatment in elderly patients with Ph+ ALL can be effective for a period of at least 6 months to almost 1 year12. Prof. Liu then outlined the local practice in China for elderly patients with Ph+ ALL mutation as she explained that these patients are often initiated onto a mild regimen consisting of either a combination of chemotherapy plus a TKI or blinatumomab plus a TKI. These findings have been substantiated in a study by Rousselot et al., (2016) that reported elderly Ph+ ALL patients treated with dasatinib combined with low-intensity chemotherapy attained a 5-year overall survival of 36%13. Prof. Takahashi, an expert hematologist from the Department of Hematology, Nephrology, and Rheumatology at the Akita University Graduate School of Medicine and Faculty of Medicine in Japan discussed the concept of chemotherapy-free regimen and the potential risk, which was highlighted by MD Anderson suggesting an increased risk of central nervous system (CNS) relapse in Ph+ ALL patients. Moreover, they have recommended incorporation of 12 prophylactic intrathecal chemotherapy (consisted mainly of methotrexate 12 mg alternating with cytarabine 100 mg given via lumbar puncture during each cycle for up to 6 cycles) to HCVAD (hyperfractionated cyclophosphamide, dexamethasone, vincristine, and doxorubicin alternating with high-dose methotrexate and cytarabine) plus a BCR::ABL1 TKI to reduce the risk of CNS relapse14,15.
In relation to the chemotherapy-free regimen, Prof. Kwong shared the local practice in Hong Kong as he clarified that patients who are on chemotherapy-free regimen still receive intrathecal prophylaxis. In addition, he explained that since ponatinib16 and blinatumomab17 are unable to cross the CNS very well, intrathecal prophylaxis is usually recommended in these patients. But is chemotherapy-free regimen applicable for patients with IKAROS deletion? Prof. Takahashi stressed that chemotherapy-free regimen is effective even for Ph+ ALL patients with IKAROS (IKZF1) deletion, however, the dasatinib-blinatumomab combination may not be as effective against the IKZF1 deletion with genetic aberrations (cyclin-dependent kinase inhibitor 2A [CDKN2A] or CDKN2B, paired box 5 [PAX5], or both [IKZF1plus])18,19 . To elaborate further, he shared the findings from a study by Dr Foà in which cohorts were divided into three groups based on their IKZF1 status: Group 1 with no IKZF1 deletion, group 2 with IKZF1 deletion and group 3 with IKZF1plus. The results from the study demonstrated that dasatinib-blinatumomab combination was less effective in Ph+ ALL patients with IKZF1plus compared to those with only IKZF1 deletion or no IKZF1 deletion19.
Despite the success of imatinib, alternative therapeutic option is often necessary, particularly for CML patients that experience disease progression while on imatinib, or had r/i to imatinib due to various side effects20. Here, Prof. Kim explained that in South Korea only imatinib and second-generation TKIs (dasatinib and nilotinib) are reimbursable front-line treatments in CML21, usually at the chronic phase. He highlighted that when patients developed CML at either an accelerated phase (AP) or with blast crisis (BC), only imatinib is used at a high dose since it is reimbursable. Regarding ponatinib, Prof. Kim explained that ponatinib is considered as a sub-line therapy, except in patients with T315I mutation, where after first-line treatment failure, ponatinib is used as a second-line treatment. When asked regarding the efficacy of asciminib against the T315I mutation, Prof. Kim advised that it is effective against T315I mutation22, however, it is not approved by the South Korean government. He added that both asciminib and ponatinib are competitive in South Korean market, nevertheless, the efficacy of ponatinib is better.
Prof. Takahashi offered his view on cases with treatment r/i as he described the European LeukemiaNet (ELN) recommendation that suggests monitoring of molecular response using quantitative polymerase chain reaction whenever possible23. In addition, he disclosed that he often uses ponatinib as a second-line treatment after first-line treatment failure in CML patients. Nonetheless, if a patient is unable to attain a MR, then addition of a second-generation TKI or a low dose of ponatinib is warranted, according to Prof. Takahashi. Interestingly, even though 40 mg twice daily (BID) asciminib is recommended and reimbursable as a third-line treatment in Japan, it is ineffective against T315I mutation as per Prof. Takahashi. Prof. Kim clarified this point further suggesting 80 mg of asciminib once daily (QD) costs around $4,000 United States dollars (USD) per month and to treat patients with T315I mutation, a 5-times the dose of asciminib is required24 which is not approved by the South Korean government.
There is a controversy about whether 3 or 6 months molecular assessment predicts the progression-free and overall survival in CML patients25. Prof. Takahashi commented that early MR is important but can be affected by several variables which include treatment tolerance, patient adherence to treatment and pharmacokinetics of the drug. He went onto suggest that he normally waits for at least a period of 6 months prior to changing the TKI and if the patient has still not achieved an early MR by then, he would change to another TKI. Here, Prof. Kim raised an important point that patients with a breakpoint cluster region-Abelson (BCR-ABL1) value of >10% at 6 months are at risk of treatment failure, therefore, warrants a switch in treatment to ponatinib. Regarding MR monitoring, Prof. Kwong shared his practice by emphasizing that he uses a time point of 3, 6 and 12 months from initiation of treatment to the treatment failure in Hong Kong and the percentage of treatment failure would dictate the treatment escalation. In addition, he also assesses the mutation profile and adjusts the treatment according to the identified mutation.
Recent studies have suggested that ponatinib causes endothelial dysfunction through its multi-kinase inhibitory properties affecting targets such as vascular endothelial growth factor receptors and can promote the expression of proatherogenic surface adhesion receptors, increasing risk of vascular occlusive events26. Prof. Kim emphasized that 25% of patients treated with ponatinib experiences ponatinib-related hypertension, which eventually requires a dose adjustment. In relation to ponatinib-related hypertension, Prof. Takahashi outlined his daily clinical practice where he often uses ponatinib at a lower dose (15 mg or 20 mg per day in females) and then gradually increases the dose to 30 mg per day according to their treatment response. An important point raised by Prof. Kwong was that he noticed a higher incidence of ponatinib-related hypertension in patients from Europe and North America, whereas only 25% of Chinese patients treated with ponatinib reported ponatinib-related hypertension. In addition, he also noticed that ponatinib-related hypertension is somehow easier to treat in Chinese patients compared to European and North American patients who often require 2-3 antihypertensives to keep blood pressure at reasonable levels; this may indicate that there is a racial difference to treatment response between the Asian and European patients.
Notably, ponatinib treatment may induce cardiovascular adverse events (CVAEs), particularly AOEs. In the 5-year follow-up of the multinational phase II Ph+ ALL and CML evaluation PACE trial, the cumulative incidence rate of AOEs was 31% in the CML-CP population, and the rate continued to increase over time27. Prof. Kim highlighted the preventative measures he takes to reduce the CV and AOE risks in his patients. He advised that he strictly monitors his patients for hypertension and uses a combination of amlodipine and an angiotensin-II-receptor antagonist to treat patients with hypertension. In addition, he requests echocardiogram (ECHO), ankle-brachial index (ABI) and ultrasound of carotid arteries at baseline and prior to patient commencing onto ponatinib. Aside from these, he also recommends his patients to undergo yearly checkups which includes a coronary and brain magnetic resonance angiography (MRA) with carotid doppler ultrasound. Similarly, Prof. Yan also implements similar measures, in addition to an electrocardiogram (ECG) in her daily clinical practice. Moreover, she recommends her patients to have carotid doppler ultrasound annually, especially those at high CV risk. Conspicuously, ponatinib has shown efficacy in CML patients who have received prior TKIs, and the risk-benefit balance should always be evaluated for individual patients28. In conclusion, there are important factors that should be considered prior to initiation of ponatinib treatment, and these include assessing the disease state, mutational status, treatment line, the reason for the change of therapy (intolerance or resistance), age of the patient, frailty and specific comorbidities29 .
References
1. Takahashi N, et al. Japanese Journal of Clinical Oncology 2024. 2. Haddad FG, et al. Hematology Am Soc Hematol Educ Program 2022; 2022(1): 213-7. 3. Lim S-N, et al. American Journal of Hematology 2015; 90(11): 1013-20. 4. Foà R, et al. Blood 2011; 118(25): 6521-8. 5. Aldoss I, et al. Blood 2023; 142(Supplement 1): 2871-. 6. Yu G, et al. Oncotarget 2017; 8(63): 107022-32. 7. Jabbour E, et al. JAMA 2024; 331(21): 1814-23. 8. Tan FH, et al. Onco Targets Ther 2019; 12: 635-45. 9. Stock W, et al. Blood 2019; 133(14): 1548-59. 10. Martinelli G, et al. Blood Adv 2022; 6(6): 1742-53. 11. Sawalha Y, et al. Int J Hematol Oncol 2018; 7(1): Ijh02. 12. Vignetti M, et al. Blood 2007; 109(9): 3676-8. 13. Rousselot P, et al. Blood 2016; 128(6): 774-82. 14. Paul S, et al. Am J Hematol 2023; 98(1): E11-e4. 15. Paul S, et al. Blood 2019; 134(Supplement_1): 3810-. 16. Abid MB, et al. British Journal of Haematology 2017; 179(3): 497-8. 17. Kopmar NE, et al. Blood 2023; 141(12): 1379-88. 18. Foà R, et al. Journal of Clinical Oncology 2024; 42(8): 881-5. 19. Foà R, et al. New England Journal of Medicine 2020; 383(17): 1613-23. 20. Wolfe HR, et al. Current Hematologic Malignancy Reports 2021; 16(5): 448-54. 21. Uhm J, et al. Blood Res 2021; 56(1): 31-7. 22. Hughes TP, et al. New England Journal of Medicine 2019; 381(24): 2315-26. 23. Hochhaus A, et al. Leukemia 2020; 34(4): 966-84. 24. Cortes JE, et al. Clinical Lymphoma Myeloma and Leukemia 2021; 21: S335-S6. 25. Chikkodi SV, et al. Clinical Lymphoma Myeloma and Leukemia 2015; 15: S114-S9. 26. Chan O, et al. Blood Adv 2020; 4(3): 530-8. 27. Caocci G, et al. Hematol Oncol 2019; 37(3): 296-302. 28. Müller MC, et al. Critical Reviews in Oncology/Hematology 2017; 120: 52-9. 29. Luciano L, et al. European Journal of Haematology 2020; 105(1): 3-15.





