

Registered Dietitian (UK) Honorary Member, Hong Kong Nutrition Association
Chronic kidney disease (CKD) is a global public health problem that an estimated 843.6 million individuals are currently affected by various stages of the disease, whereas the prevalence of CKD is increasing, and Hong Kong is no exception1. Yet, the public awareness of CKD in Hong Kong, including the risk factors and dietary requirements of patients with the disease, is suboptimal. While protein restriction has long been used among CKD patients to delay the decline in kidney function, rigid dietary control is hard to follow in the long term and may increase the risks of malnutrition2. In this regard, supplementation with ketoanalogues (KA) coupled with a low-protein diet (KA-LPD) would reduce the risks of protein deficiency without increasing nitrogen burden and preserve kidney function in CKD patients. To uncover the dietary concerns in CKD and the clinical performance of KA, Dr. Terry Ting Ho-yan, a registered dietitian (UK), was invited to share his insights into optimising dietary management in CKD.
The Major Risk Factors of CKD
It has been reported that the most important risk factors for CKD include age, diabetes mellitus (DM), hypertension, and obesity3. Particularly, suboptimal controlled DM can lead to the development and progression of CKD that 29% to 38% of diabetic individuals develop CKD after a median followup of 15 years4. Besides DM, a local study by Wan et al. (2019) suggested that each 10 mmHg increase in systolic blood pressure (SBP) was associated with a 22% higher risk of CKD5. “It is very common that younger people who are not aware of their blood pressure develop CKD eventually,” as per Dr. Ting. Similarly, a previous study reported that each rise in 1 unit of body-mass index (BMI) confers a 20% increase in the risk of CKD over 20 years6. Thus, proper control of glycaemic levels, blood pressure, and weight is paramount in reducing the risk of CKD.
Dr. Ting addressed that lifestyle factors, such as tobacco smoking and alcohol consumption, also increase the risk of CKD. Remarkably, he added that, while certain diseases and injuries would result in kidney impairment, improper use of medications, including Chinese herbal medicine and nonsteroidal anti-inflammatory drugs (NSAIDs), can also lead to CKD. “Consulting healthcare professionals before taking medications is very important to prevent treatment-related adverse events,” Dr. Ting advised.
The Progressive Damage to Kidneys
“Stage 1 to 3 CKD is usually asymptomatic and is not easy for patients to realise the disease unless regular blood tests are conducted,” Dr. Ting noted. CKD is defined as an abnormality in kidney structure or function persisting for over 3 months. According to the KDIGO classification, CKD is classified based on the estimated glomerular filtration rate (eGFR), albuminuria, and cause of CKD. Briefly, stage 1 (eGFR of >90 ml/min/1.73m2) represents a normal kidney function, whereas stage 2 (eGFRof 60-89 ml/min/1.73m2) indicates a mild decrease in kidney function. When eGFR drops to 45-59 ml/min/1.73m2 (stage 3a), mild-to-moderate decline in kidney function is indicated7. Dr. Ting outlined that increased serum level of creatinine becomes notable in stage 3a, whereas complications, such as imbalance of electrolytes and oedema, may be observed if eGFR further reduces to 30-44 ml/min/1.73m2 (stage 3b)7. “At this stage (stage 3b), dietary restriction is needed to control blood glucose and pressure,” Dr. Ting addressed.
Notably, an eGFR of 15-29 ml/min/1.73m2 is classified as stage IV CKD, at which the kidney function is severely decreased with significant complications requiring more stringent symptom control measures. Eventually, an eGFR <15 ml/min/1.73m2 indicated end-stage renal disease (ESRD), and renal replacement therapy is needed. A summary of KDIGO CKD definition and prognosis is illustrated in Figure 17.

Figure 1. KDIGO Definition and prognosis of CKD by GFR and albuminuria categories, green: low risk; Yellow: moderately increased risk; orange: high risk; red: very high risk7
Controlling Protein Intake – Protein Quality Matters
Given that protein restriction helps reduce the disturbances characteristic of uraemia and diminishes the ill effects of hyperphosphatemia, metabolic acidosis, hyperkalaemia, and other electrolyte disorders, the Kidney Disease Outcomes Quality Initiative (KDOQI) Clinical Practice Guidelines advocate considering a LPD for patients with CKD. For instance, a controlled LPD providing 0.55-0.60 g protein/kg body weight/ day or a very low-protein diet (VLPD) providing 0.28–0.43 g protein/kg body weight/day with additional keto acid/amino acid analogues is recommended for CKD patients who are not on dialysis and without diabetes8.
Regarding protein intake, Dr. Ting emphasised that, apart from common protein-rich food, such as eggs, meat, fish, milk, and bean products, there are low-quality proteins in carbohydraterich food, such as wheat gluten. Protein quality refers to the availability of amino acids that it supplies9. Hence, it is crucial to consider the low-quality protein contents in designing recipes for LPD. “Even though the meat consumption is low, a large amount of rice intake will increase the total protein intake,” Dr. Ting noted. The protein quality represented by the Protein Digestibility Corrected Amino Acid Score (PDCAAS) of some food substances is shown in Figure 29.

Figure 2. Protein quality of common food products9, PDCAAS: Protein Digestibility Corrected Amino Acid Score
Implementing LPD involves significant changes in dietary habits, which may be difficult practically. Thus, Dr. Ting suggested that a stepwise approach can be considered. Furthermore, he highlighted that referral to dietitians for advice on dietary planning is desirable for CKD patients with stage 3 disease or above.
The Roles of KAs in Optimising LPD
If the protein intake is well-restricted, KAs of essential amino acids (EAAs) can utilise circulating amino groups to transfer themselves into EAAs through the transamination effect. Hence, supplementation with KAs can reduce the risks of protein deficiency without increasing the nitrogen burden for patients on LPD or VLPD2.
While clinical guidelines recommend supplementation with KAs for CKD patients8, Dr. Ting stated that KA-LPD has long been used in local clinical practice. “KAs allow the reduction of dietary protein intake to an average of about 0.6 or even 0.5 g protein/kg body weight/day. While reducing the workload of the kidneys, KAs slow down the progression of kidney impairment and the effect of uraemia,” Dr. Ting said. He added that, based on his clinical observation, no clinically significant adverse events are associated with the use of KAs. However, he reminded that consulting healthcare professionals before using KAs is essential for achieving optimal outcomes.
The Clinical Benefits of KAs
The clinical benefits of KA-LPD have been demonstrated in various clinical trials. The retrospective cohort study involving 1,042 pre-dialysis CKD patients (stage 3-5) received KA-LPD by Ariyanopparut et al. (2022) revealed that, after a median follow-up of 32.9 months, patients received KA-LPD had a significantly lower risk of kidney function decline (hazard ratio [HR]: 0.13, p <0.001) and dialysis initiation (HR: 0.24, p <0.001, Figure 3) than those received LPD alone after adjusting for confounders10.
Figure 3. Kaplan–Meier estimates of primary composite endpoint Figure 4. Cumulative event rate of ESRD requiring dialysis2 10, defined as an annual eGFR decline of more than 5 ml/min/1.73 m2 or long-term dialysis initiation
On the other hand, a retrospective analysis including 541 patients with stage 4 CKD received KA-LPD by Yen et al. (2022) demonstrated that continuation of KA-LPD (n=303) exhibited a significantly lower incidence of new-onset ESRD requiring maintenance dialysis (6.8% vs. 10.4%, HR: 0.62, p =0.023) than those discontinued supplementation with KAs (n=238, Figure 4)2. Based on the clinical studies above, initiating KA-LPD and continuing the KA supplementation would reduce the short-term risk of commencing dialysis.
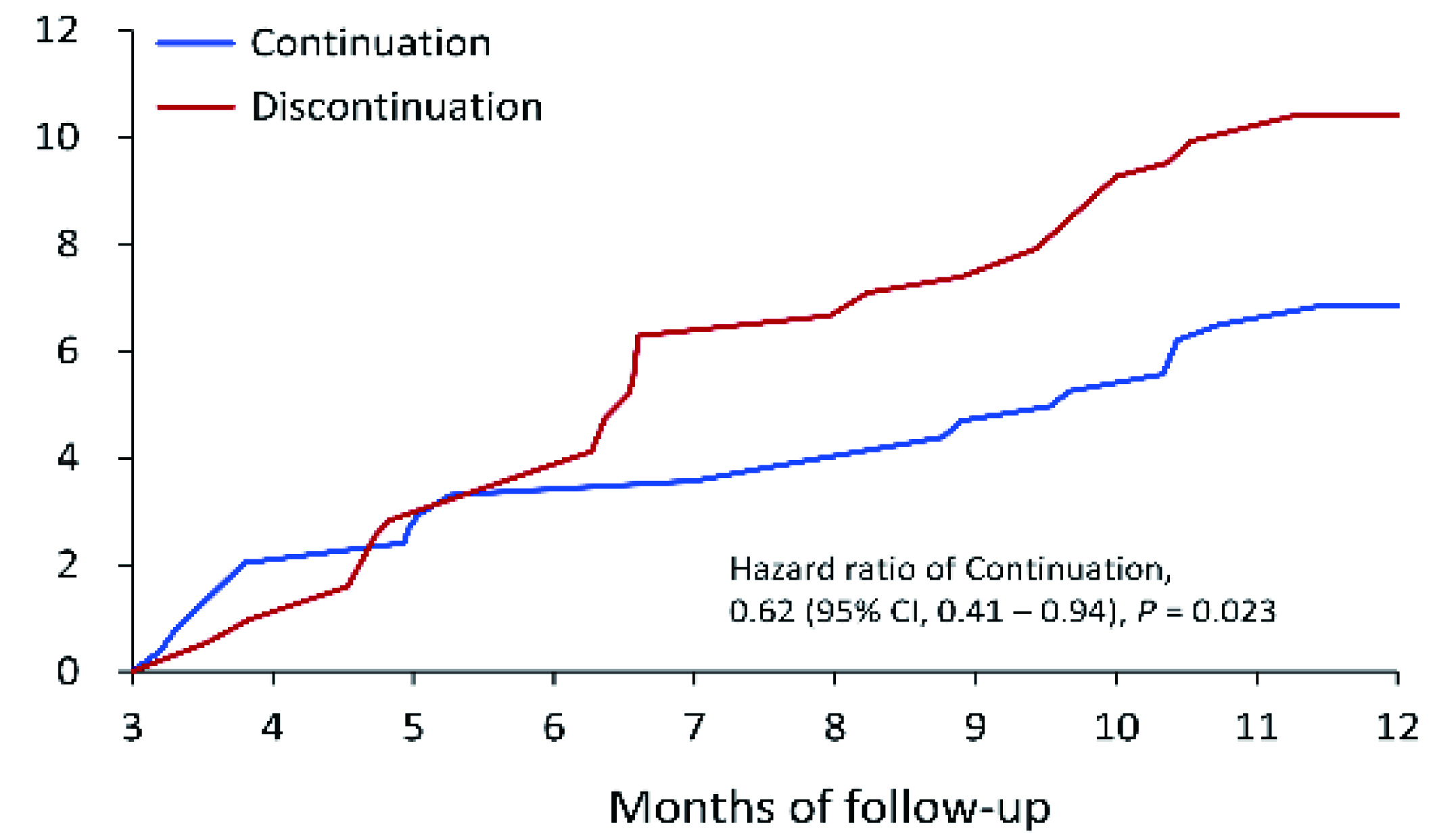
Figure 4. Cumulative event rate of ESRD requiring dialysis2
Apart from pre-dialysis CKD patients, Dr. Ting noted that KA supplementation can be prescribed for ESRD patients to preserve their residual kidney functions. “In patients receiving dialysis treatment, KA supplementation helps maintain the patient’s residual kidney function and, hence, increases the efficacy of dialysis,” he highlighted. Moreover, while KA supplementation is prescribed for patients with stage 5 CKD, there are studies investigating the clinical outcomes of early use of KA-LPD11.
Getting the Right Advice from the Right Sources
Based on the published clinical data, KA-LPD is effective in preserving kidney functions for CKD patients, with no clinically significant adverse events observed. Nonetheless, Dr. Ting addressed that many CKD patients adopted a too stringent dietary control, which potentially leads to suboptimal outcomes. “Although control of dietary potassium and phosphorus intake is important, the level of control varies according to the stage of the disease, but not the lower, the better,” he stated. Besides, he pointed out that patients are nowadays flooded with dietary information from the internet, whereas a substantial proportion of them may not be accurate. In particular, there are opinions advocating the keto diet and low-carbon diet, which may not be suitable for CKD patients.
As a final remark, Dr. Ting stressed that consulting nephrologists and dietitians is essential in planning the diet for CKD patients. In particular, while many CKD patients concern about the impact of protein restriction on their daily food options, dietitians play a key role in designing suitable recipes for them, balancing the low-protein requirements and patient’s enjoyment of food.
References
1. KKovesdy CP. Kidney Int Suppl (2011) 2022; 12: 7. 2. Yen et al. Nutrients 2022; 14. DOI:10.3390/NU14194020/S1. 3. Mallamaci and Tripepi. Journal of Clinical Medicine 2024, Vol 13, Page 678 2024;
13: 678. 4. Ooi et al. Diabetes Metab J 2024; 48: 196. 5. Wan et al. Hypertension 2019; 74: 331–40. 6. Than et al. Adv Biomark Sci Technol 2020; 2: 24–34. 7. Chen et al. JAMA 2019; 322: 1294. 8. Ikizler et al. American Journal of Kidney Diseases 2020; 76: S1–107. 9. Hoffman and Falvo. J Sports Sci Med 2004; 3: 118. 10. Ariyanopparut et al. Scientific Reports 2023 13:1 2023; 13: 1–10. 11. Li et al. Nutrients 2019; 11. DOI:10.3390/NU11050957.





